INTRODUCTION TO VISUAL PROSTHETICS by Eduardo Fernandez and Richard Normann
“The best and most beautiful things in the world cannot be seen or even touched – they must be felt with the heart ” Hellen Keller.
1. Introduction.
Loss of vision affects millions of people worldwide and poses extraordinary challenges to individuals in our society that rely heavily on sight. Although in recent years the techniques of molecular genetics have led to a rapid identification of a great number of genes involved in visual diseases (see https://sph.uth.edu/Retnet/sum-dis.htm for an update of genes and loci causing retinal diseases), the nervous system once damaged is capable of little functional regeneration and currently there is no effective treatment for many patients who are visually handicapped as a result of degeneration or damage to: 1) the retina, 2) the optic nerve, or 3) the visual parts of the brain.
While pharmacological interventions provide therapeutic solutions to many physiological problems, a pharmacological approach to the mechanisms of blindness has not been discovered. Furthermore, there remains the pervasive question as to how these molecular approaches will actually restore functional vision after it is completely lost in a given individual. Therefore, there are compelling reasons to pursue the development of sophisticated microelectronic prostheses as a viable rehabilitative and therapeutic options to substitute, and ultimately, restore limited, but useful sight. Such assistive devices have already allowed thousands of deaf patients to hear sounds and acquire language abilities and the same hope exists in the field of visual neurorehabilitation.
Essentially all visual prosthesis efforts share a common principle of providing focal electrical stimulation to intact visual structures (including the retina, optic nerve, lateral geniculate nucleus (LGN) and occipital visual cortex) evoking the sensation of discrete points of light called phosphenes. Figure 1 shows the possible perception of phosphenes generated by stimulating simultaneously 3 electrodes arranged as a triangle.
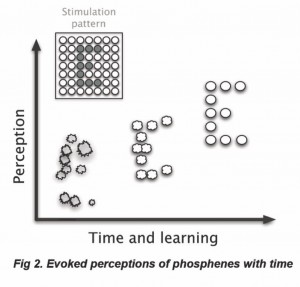
It is expected that the neural plasticity of the visual system can contribute to an ever-improving correlation between the physical world and evoked phosphenes. Figure 2 shows an example. Immediately after implantation of the evoked phosphenes that are likely to induce a poor perception of an object (the letter “E” in this example). However, appropriate learning and rehabilitation strategies will contribute to provide concordant perceptions.
2. The History Of Neural Stimulation.
Electrical phenomena and some of the effects of electricity upon man were known in ancient times, but it was not until the eighteenth century that a connection was made between electricity and the neural system. In 1791 Luigi Galvani, an Italian physician, observed that dissimilar metals, attached to a frog’s leg and connected together can induce the frog’s skeletal muscles to contract (Galvani 1791).
These observations lead to the conclusion that nerves and muscles could produce electricity and utilize it to function. Subsequent studies led to a further understanding of the nerve impulse and synaptic transmission and to the development of new therapies based on the application of electrical fields.
The first and most successful example of a neural stimulation device is the cardiac pacemaker that has become a standard therapeutic approach to improve cardiac function in millions of patients. Technology partially achieved during the development of the pacemaker has been used successfully for rehabilitation of sensory and/or motor functions in patients with neurological diseases. Thus deep brain stimulators have been implanted successfully in patients for pain management and for control of motor disorders such as Parkinson’s disease (Hunter, Yoshino et al. 2004; Pena, Bowsher et al. 2004; Stieglitz, Schuettler et al. 2004), and cochlear implants are being used for restoring auditory function (Brors and Bodmer 2004; Chatelin, Kim et al. 2004; Cohen 2004; Balkany, Hodges et al. 2005; Chang 2005). Moreover advances in artificial limbs and brain-machine interfaces are now providing hope of increased mobility and independence for amputees and paralyzed patients (Donoghue 2002; Nicolelis 2003; Donoghue, Nurmikko et al. 2004; Carmena, Lebedev et al. 2005; Hochberg, Serruya et al. 2006) and there is preliminary data showing that electrophysiological methods can be used to extract neural information about the volitional intent of the subjects to move their distal musculature and then translate these signals into models that are able to control external devices (Barbeau, McCrea et al. 1999; Donoghue 2002; Nicolelis and Chapin 2002; Serruya, Hatsopoulos et al. 2002; Paninski, Fellows et al. 2004; Patil, Carmena et al. 2004; Sanchez, Carmena et al. 2004; Hochberg, Serruya et al. 2006). As more and more patients have benefited from this approach, the interest in neural interfaces and visual prostheses has grown significantly.
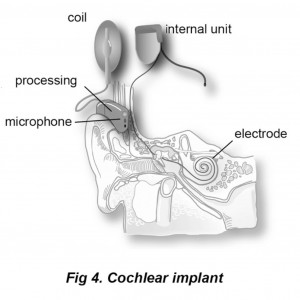
In terms of restoring lost sensory function, cochlear implant research has arguably been the most successful and has been translated into a viable therapeutic option for many profoundly deaf patients (Loeb 1990; McDermott 2004; Pena, Bowsher et al. 2004; Bouccara, Avan et al. 2005). Simply explained, a wire electrode bundle is surgically inserted into the inner ear thus serving as an electronic replacement for damaged hair cells. A microphone and speech processor picks up and decodes sounds from the environment and the coded signals drive, in turn, the electrodes that stimulate the nerve fibers of the cochlea, and create the sensation of hearing. In conjunction with intense rehabilitation programs that are tailored to the individual, deaf individuals can learn to comprehend and in some cases, even acquire speech. The same hope exists in the field of visual prostheses.
This field is still in its infancy but is experiencing great interest in both academic and commercial sectors. The possibility of merging different technologies and focus them on a common target is allowing us to develop new systems, devices and interfaces to the nervous system with greater complexity than could have been imagined only a few decades ago. Further, our understanding of the function and dysfunction of the nervous system has improved to the point that, in many cases, researchers have suggested plausible interventional routes whereby neuroprosthetic systems can be expected to be efficacious.
3. The Case For Artificial Vision.
The possibility to restore visual perceptions in blind individuals has a long history in biomedical engineering. A German neurosurgeon, Forester, was the first to expose the human occipital pole under local anesthesia and to electrically stimulate it. In 1929 he noted that electrical stimulation induced the perception of points of light, called phosphenes and usually described as ‘stars in the sky’, ‘clouds’ and ‘pinwheels’, that were dependent on where the stimulation probe was placed. These findings together with the studies of Wilder Penfield and co-workers during the course of neurosurgical interventions for the treatment of epilepsy (Penfield and Rasmussen 1950; Penfield and Jaspers 1974) established the physiological basis for the present efforts to develop a visual prosthesis for the blind.
Subsequent experiments by the group of Giles Brindley in England (Brindley and Lewin 1968; Brindley, Donaldson et al. 1972; Brindley 1982), William Dobelle at the University of Utah (Dobelle and Mladejovsky 1974; Dobelle, Mladejovsky et al. 1974; Dobelle, Mladejovsky et al. 1976; Dobelle 2000), Pollen (Pollen 1975) and others (Bak, Girvin et al. 1990) showed that simultaneous stimulation via arrays of electrodes placed on the surface of the brain allowed blind volunteers to recognize simple patterns, including letters and Braille characters.
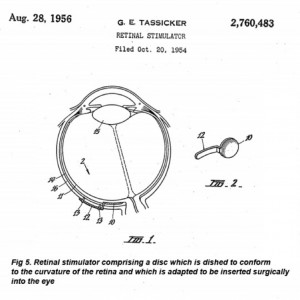
The results of these studies supported the premise that patterned electrical stimulation of the visual cortex can evoke patterned percepts. However, these early efforts did not culminate in the restoration of a useful visual sense. Problems with this early work were associated mainly with the large surface electrodes that were used to evoke phosphenes. Such large surface area electrodes required relatively high electrical currents to evoke phosphenes and when multiple electrodes were stimulated, these large currents could interact in a non-linear fashion, evoking phosphenes with unpredictable spatial properties. Furthermore they also found that a visual prosthesis based on relatively large surface electrodes implanted subdurally would have limited usefulness because of occasional elicitation of pain due to meningeal or scalp stimulation, and also the risk of inducing epileptic seizures. This body of work suggests that relatively large surface electrodes may not be a tenable approach to stimulation of the visual cortex, and led a number of investigators to develop a visual prosthesis that could be implanted directly into the retina, the brain or other parts of the visual pathways. One of the pioneers in this field was Tassicker, an Australian researcher who in 1956 was the first to patent a method of implanting a light-sensitive selenium cell in a blind person’s retina to transiently restore the patient’s ability to perceive the sensation of light (Tassicker 1956; Zrenner 2002).
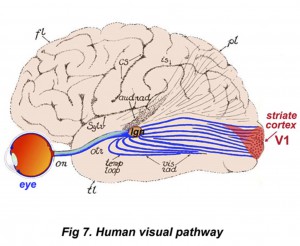
4. Visual Pathways: From Real Vision To Visual Neuroprostheses.
The concept of artificially producing a visual sense in blind individuals is founded on our present understanding of the structure of the mammalian visual system, its processing elements, and the relationship between electrical stimulation of any part of the visual pathways and the resulting visual sensations (Maynard 2001).
Our entire experience of the external visual world derives from the concerted activity of a restricted number of retinal ganglion cells (Figure 6).
Figure 6. Cross-section of the human retina. Photoreceptors are at the top and ganglion cells at the bottom. Light enters the retina from the bottom and travels through the thickness of the retina to stimulate the photoreceptors at the top in this view. (Drawing courtesy of Helga Kolb)
These retinal ganglion cells have to send their action potentials through the “bottleneck” optic nerve to the brain (Figure 6).
The retina is essentially a piece of brain whose 150 million photoreceptors get direct stimulation from the outside world’s lights and images. Visual input to the retina consists of a stream of photons, which can be unequivocally quantified in space and time. The retina performs spatial, temporal, and chromatic processing of visual information and converts it into a compact “digital” format composed of neural impulses (Kolb, Fernandez et al. 2005). In addition, starting at the retinal level, vision is an active process and eye movements are essential for information processing (Wilke, Thiel et al. 2001; Greschner, Bongard et al. 2002). Thus our entire experience of the external visual world derives from the concerted activity of a restricted number of retinal ganglion cells, which have to send their information, via the one million optic nerve fibers, to higher visual centers. The representation has to be unequivocal and fast (within a few hundreds of milliseconds), in order to ensure object recognition for any single stimulus presentation (Bialek, Rieke et al. 1991; de Ruyter van Steveninck, Lewen et al. 1997; Smirnakis, Berry et al. 1997). Therefore the question of how the information about the external world is compressed in the retina, and how this compressed representation is encoded in spike trains is an important challenge for the success of any visual prosthesis.
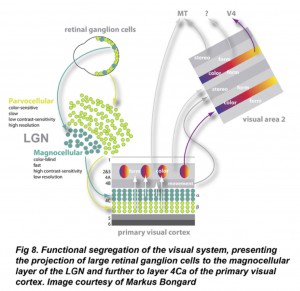
The next processing center is the lateral geniculate nucleus (LGN), which begins to integrate information from both eyes into a binocular representation of visual space. Once retinal signals arrive within the LGN, they are in a position to be influenced by a wide variety of other inputs as well as the complex feedforward and feedback circuits to and from higher visual centers. These inputs include not only visual information, but also nonvisual information that comes from cortical areas, superior colliculus, pretectum, parabigeminal nucleus, reticular nucleus and other brain stem pathways (Sherman and Guillery 2001). The number of synapses provided by these non-retinal inputs greatly outnumbers the number of the parallel pathways coming from the retina, but their function is not fully understood.
The LGN neurons mainly project to the primary visual cortex (also known as the striate cortex or V1), which was the first cortical area clearly identified in the human brain by Gennari in 1782 (Gross 1997). Area V1 is located at the back of the head, in the occipital lobe, and from here the information is distributed to a number of higher cortical centers for further processing (Grill-Spector and Malach 2004). The main point, as stated by Hubel and Wiesel, is that the primary visual cortex is in no sense the end of the visual path (Hubel and Wiesel 1977; Hubel and Wiesel 1990). It is just one stage, probably an early one in terms of the degree of abstraction of the visual information processing (Hubel and Wiesel 1977; Dougherty, Koch et al. 2003; Wandell and Wade 2003; Schmolesky 2005).
As blindness results from an interruption in the normal flow of signals along the visual pathways, a visual prosthesis has to excite the neurons of the pathway at some point after the damage site (Normann, Maynard et al. 1996; Fernandez, Pelayo et al. 2005). The only requirement is that the device should contact still functioning neural elements.
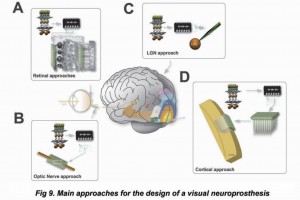
5. Current Approaches To A Visual Prosthesis.
Figure 9 shows the main approaches for the design of a visual neuroprosthesis.
Since retinal diseases frequently reduce visual acuity and result in non-curable blindness, several groups worldwide are working on the development of different prostheses designed to interact with the remaining healthy retina (Eckmiller 1997; Walter, Szurman et al. 1999; Zrenner 2002; Humayun, Weiland et al. 2003; Rizzo, Wyatt et al. 2003), optic nerve (Delbeke, Pins et al. 2001; Veraart, Wanet-Defalque et al. 2003) and LGN (Pezaris and Reid 2007; Pezaris and Eskandar 2009; Jeffries, Killian et al. 2014). However the output neurons of the eye, the ganglion cells, often degenerate in many retinal blindnesses (Jones, Watt et al. 2003; Marc and Jones 2003; Jones and Marc 2005; Jones, Kondo et al. 2012; Marc, Pfeiffer et al. 2014), and therefore a retinal, optic nerve or LGN prosthesis would not always be helpful. This extensive degeneration usually spares the neurons in the higher visual regions of the brain, which suggest the enormous potential of a cortical prosthesis designed to stimulate cortical neurons (Normann, Maynard et al. 1996; Schmidt, Bak et al. 1996; Normann, Greger et al. 2009; Fernandez, Greger et al. 2014). Due to complexity of the interconnectivity and the receptive field characteristics, visual areas beyond V1 have not been proposed as sites for vision prostheses.
Retinal Stimulation
Retinal prostheses are being developed to apply electrical stimulation to the retina in order to restore vision in patients with age-related macular degeneration and retinitis pigmentosa. More than five groups are already pursuing human clinical trials of various retinal prostheses (Dorn, Ahuja et al. 2013; Zrenner 2013; Kitiratschky, Stingl et al. 2015; Stingl, Bartz-Schmidt et al. 2015).
There are significant advantages to the retinal approach as it is less invasive and the implants are lower in the visual pathways which means that they are closer to the photoreceptors and have more opportunities for natural processing of the visual information. Further, the retinal approach has the advantage that no camera is required to encode the visual world: the optics of the eye provide this function. However there are also some problems related to encapsulation of the device, fixation of the implants, power dissipation and the production of sufficient currents (Normann 2007; Normann, Greger et al. 2009). Furthermore for a retinal visual prosthesis to work, there must be a population of functional retinal ganglion cells, and therefore these systems are not capable of treating very advanced retinal degenerations (Dagnelie 2006; Dowling 2009).
Currently there are mainly two kinds of retinal implants under development: subretinal and epiretinal. Furthermore some groups are studying the efficacy of the suprachoroidal-transretinal stimulation to generate focal excitation in retinal ganglion cells (Nakauchi, Fujikado et al. 2007; Nishida, Kamei et al. 2010; Nayagam, Williams et al. 2014; Schmid, Fink et al. 2014; Shivdasani, Sinclair et al. 2014; Yan, Sui et al. 2015). The latest experimental photostimulation approach involves re-engineering retinal ganglion cells or bipolar cells to become light sensitive by incorporation of artificial opsins (Degenaar, Grossman et al. 2009; Busskamp, Picaud et al. 2012; Roska, Busskamp et al. 2013; Sahel and Roska 2013; Trenholm and Roska 2014). This novel approach is based on laboratory studies in fish or mouse and are nowhere near ready for human application.
Subretinal Implants
As some of the neural retina, with the exception of the photoreceptors layer, is preserved in both age-related macular degeneration and retinitis pigmentosa a reasonable approach to the restoration of vision under these pathologies is the replication of the function of the photoreceptors (Rizzo, Wyatt et al. 2001; Zrenner 2002; Zrenner 2013; Kitiratschky, Stingl et al. 2015; Stingl, Bartz-Schmidt et al. 2015). One of the main advantages of this location is that the remaining retinal cells in the outer plexiform layer (vertically running bipolar cells and horizontally oriented horizontal cells) may be utilized to stimulate the amacrine and ganglion cells directly.
In a general sense all research groups that have focused on the subretinal approach have proposed a similar method. Thousands of light-sensitive microphotodiodes equipped with microelectrodes are assembled on a very thin plate and placed in the subretinal space. The subretinal device or microphotodiode array (MPDA) is implanted between the pigment epithelial layer and the outer layer of the retina, which contains the photoreceptors. Light falling on the retina generates graded currents in the MPDA that stimulate the dendrites of the bipolar cells in the outer plexiform layer to induce a visual sensation.
The schema is tantalizingly simple. Furthermore the photodiodes can be tailored to provide either positive or negative current in response to illumination, with the intention of mimicking the function of ON and OFF bipolar cells. However the success of this approach depends upon three assumptions. First, the bipolar cells of the dysfunctional retinal should persist and function in a somewhat physiologically normal manner. Second, the MPDA must generate enough current under normal illumination levels to stimulate the remnant bipolar cells. Third, the electrodes can be placed in close enough proximity to the bipolar cells. Encouraging progress has been reported in each of these areas.
Epiretinal Implants
As an alternative approach to stimulate the retina, other research groups have proposed placing the stimulating electrodes in close proximity to the retinal ganglion cells. This approach attempts to stimulate the remaining retinal neurons of patients who are blind from end-stage photoreceptor diseases and relies on significant numbers or retinal ganglion cells surviving in pathologies such as age-related macular degeneration and retinitis pigmentosa (Stone, Barlow et al. 1992; Santos, Humayun et al. 1997).
Both the epiretinal and subretinal approaches imply the use of implanted multielectrode arrays (MEAs) and transcutaneous telemetry to transfer data and power to the implanted MEAs. To date, a number of experiments performed in sighted and blind human subjects have demonstrated the potential for epiretinal electrical stimulation to provide patterned visual perception. One such device, the Argus II epiretinal implant developed by Second Sight (Sylmar, California, USA) has already obtained regulatory approval for marketing in Europe and the United States and has show improved reading and motion detection in several patients (Humayun, Dorn et al. 2012; Dorn, Ahuja et al. 2013; Stronks and Dagnelie 2014). There are, however, issues relevant to the epiretinal approach that need to be solved. The epiretinal devices have to be firmly affixed in place in order to efficiently stimulate the retina and provide a consistent visual perception. Other issues of concern are power dissipation, cross-talk between electrodes, and the long-term viability of the tissues under the implant.
LGN Stimulation
The lateral geniculate nucleus (LGN) is the primary processing center for visual information, meaning that the neural signals encoding visual information have not yet been extensively processed. The LGN receives information directly from the retinal ganglion cells and projects mainly to the primary visual cortex. The LGN holds promise as a target for electrical stimulation for artificial visual percepts since it has been demonstrated that LGN microstimulation in monkeys produces predictable visual percepts (Pezaris and Reid 2007; Pezaris and Eskandar 2009; Jeffries, Killian et al. 2014). Therefore, a visual prosthesis based on LGN stimulation could be helpful to restore sight to those who have become blind because of trauma to the eye or diseases such as glaucoma, macular degeneration, or retinitis pigmentosa.
One possible disadvantage of this approach is the limited number of electrodes that can be inserted in the LGN and hence the limited visual resolution capable with this approach. Moreover, the neurons in the LGN are too close together to be stimulated individually, which would be important in trying to reproduce natural vision. Furthermore most of the projections to the LGN are from primary visual cortex making it difficult to create complex visual percepts.
Intracortical Stimulation
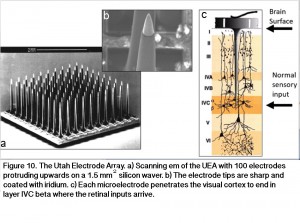
If primary visual cortex neurons can be stimulated with visual information in a format somewhat similar to the way they were stimulated before the onset of blindness, a blind individual could use this stimulation to extract information about the physical world around him/her (Normann, Maynard et al. 1996; Fernandez, Pelayo et al. 2005; Normann, Greger et al. 2009; Fernandez, Greger et al. 2014). Thus in more recent years an intracortical approach with penetrating microelectrodes has been introduced. Penetrating electrodes can activate populations of neurons with greater spatial specificity and lower levels of stimulation than is possible with larger electrodes on the surface of the brain as in the original Brindley and Dobelle experiments. The Utah Electrode Array (Campbell, Jones et al. 1991) is an example of such a penetrating microelectrode array that could be used to stimulate many neurons in the visual cortex (Figure 10). The active regions of the electrodes in this electrode array are coated with iridium oxide to facilitate the transfer of electrodes flowing in the conducting silicon from which the array is made, and the size of these active regions is on the order of 50 µm. Such small electrodes abut small populations of cortical neurons, and electrical currents on the order of 10-30 µA can excite these neurons (this is three orders of magnitude smaller than the currents required with large surface electrodes). The electrodes are 1.5 mm in length and when implanted in visual cortex they reach down to layer 4, the layer of the cortex that receives normal input from the retina (Figure 10). In order to restore useful vision, visual cortex would have to be tiled with from 5-10 UEAs (Cha, Horch et al. 1992).
In 1966, Schmidt et al. (Schmidt, Bak et al. 1996) described the implantation of 38 floating microelectrodes within the right visual cortex of a 42-year-old woman who had been blind over 22 years. 34 of the microelectrodes were able to elicit phosphenes for a period of 4 months, and most of the microelectrodes had stimulation thresholds below 25 mA. Unfortunately these microelectrodes were not well suited for long-term application. Thus, due to the breakage of lead wires early in the experiment, only limited tests could be done to evaluate recognition of simple patterns of stimulation. Nevertheless, taken as a whole the results suggested that passing electrical currents through an array of electrodes inserted into an appropriate location in the visual pathway, is able to produce the perception of phosphenes, and that these phosphenes could be appropriate for restoring some limited but useful sense of vision to the profoundly blind. This intracortical approach requires penetrating electrodes with exposed tips located in layer 4 and with sizes that are of the same order of magnitude as the neurons that are intended to be stimulated.
This approach would be the most viable treatment for complete blindness caused by glaucoma, optic atrophy or diseases of the central visual pathways, such as brain injuries or stroke. Several groups worldwide are presently working on it (Bradley, Troyk et al. 2005; Fernandez, Pelayo et al. 2005; Normann, Greger et al. 2009; Kane, Cogan et al. 2013; Fernandez, Greger et al. 2014; Lewis, Ackland et al. 2015).
6. Engineering A Visual Neuroprostheses.
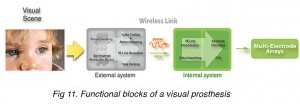
Regardless of the differences in the approaches, all vision prosthesis (with the exception of subretinal prostheses and the photostimulation approach) share a common set of components (Maynard 2001) which are illustrated in Figure 9. One or two cameras provide image acquisition. These visual signals are then processed by a bio-inspired retina-like encoder in order to transform the visual world in front of a blind individual into electrical signals. This first stage performs a multichannel spatio-temporal filtering, to extract and enhance the most relevant features of the scene and also to re-encode this information into a neuromorphic stream of electrode addresses (Pelayo, Romero et al. 2004). The second stage serializes the information and transmits it through a radio-frequency link to the implanted device. This telemetric transmission provides a wireless transfer of power and data to the internal system. The implanted electronics package must decode the signals, identify the target electrodes and generate the stimulation signals applied using multiple microelectrodes.
Most of the devices include a bio-inspired visual information block (artificial retina) to extract and enhance the most relevant features of the visual scene. A coding block that translates the retina-like output into pulses modulated with different cadences and temporal lags and whose projection onto the electrode matrix must be fully configurable. A radio frequency block provides a wireless transfer of power and data to the implanted electronics package which must decode the signals, identify the target electrodes and conform the voltage shape (D/A converter) of the final waveforms to be applied to the electrodes located near the target neurons (see Fig 11).
These transformations must take into account many issues that are normally performed by the retina. For example, sampling across the retina is not uniform and therefore retinotopic gradients and magnification factors have to be introduced to match image representation. On the other hand it is clear that several streams of information are processed in parallel from any retinal point by several dozens of interneuronal subtypes before contrast, brightness, orientation movement and color are finally coded as modulation of ganglion cell action potential series (Kolb, Nelson et al. 2001). While chromatic information is not of utmost priority, a differential characterization will nevertheless be required when designing “achromatic” processing modules for basic representation of image components. The goal of developing such a bio-inspired retinal encoder is not to simply record a high resolution image, but to transmit visual information in a meaningful way to the appropriate site(s) in the retina, optic nerve, LGN or brain.
Although it is not the purpose of this chapter to present a detailed study of the problem of coding/decoding of retinal images by ensembles of retinal ganglion cells, increasing evidence suggests that the retina and the brain utilize distributed codes that can only be analyzed by simultaneously recording activity of multiple neurons (Fernandez, Ferrandez et al. 2000; Normann, Warren et al. 2001; Warren, Fernandez et al. 2001; Schnitzer and Meister 2003). Far from a simple transducer of light into electrical neural impulses, the retina performs a locally-computed spatio-temporal contrast enhancement function, and a very efficient compression of visual information. These tasks are essential to provide a high adaptation capability to very different lighting conditions, high noise immunity, and to efficiently communicate the visual information by means of a limited number of optic nerve fibers. Thus, our entire experience of the external visual world derives from the concerted activity of a limited number of ganglion cells in the retinal output layer. They have to represent all the features of objects in the visual world, namely their color, intensity, shape, movement, and the changes in these features in time. This representation has to be unequivocal and fast (within a few hundreds of milliseconds) in order to ensure object recognition for any single stimulus presentation.
The development of a bio-inspired visual encoder poses therefore an information processing challenge without parallel in neuroscience, computer science or communication technologies. Consequently, several researches are developing complex retina encoders for visual implants (Eckmiller, Neumann et al. 2005; Morillas, Romero et al. 2007).
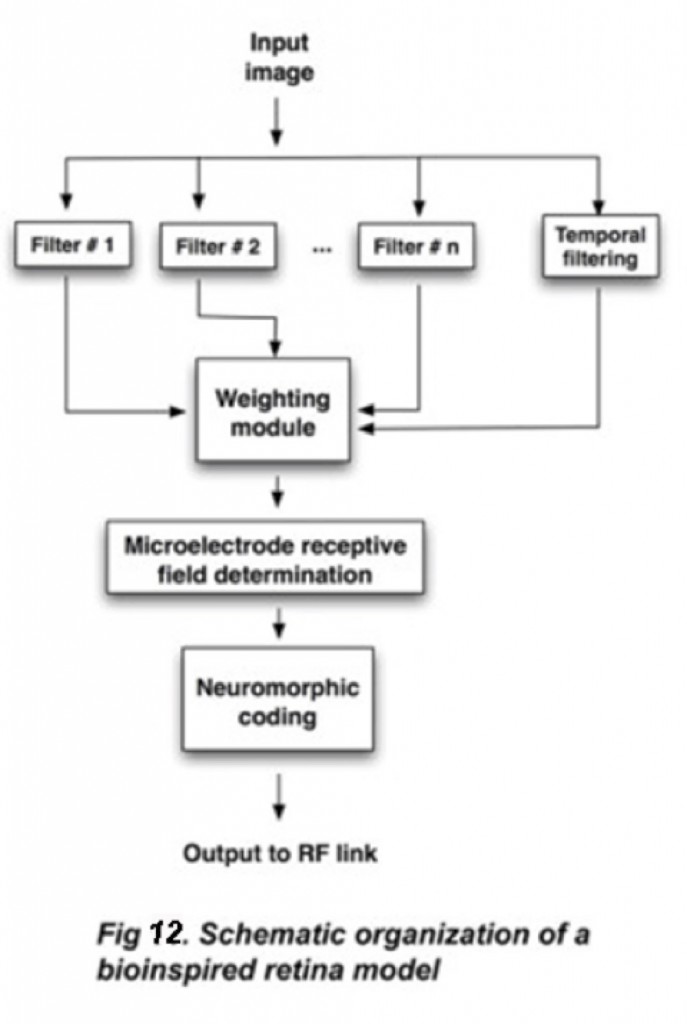
Figure 12 summarizes the basic architecture of a typical bio-inspired retinal model. It is based on information about the photoreceptor distribution within the human/primate retina and on electrophysiological recordings from populations of retinal ganglion cells. Through a set of parametrized filters and functions, that simulate the complex operations of the neural retina, a portable model is obtained that can be easily translated into a hardware description for automatic synthesis using the appropriate tools.
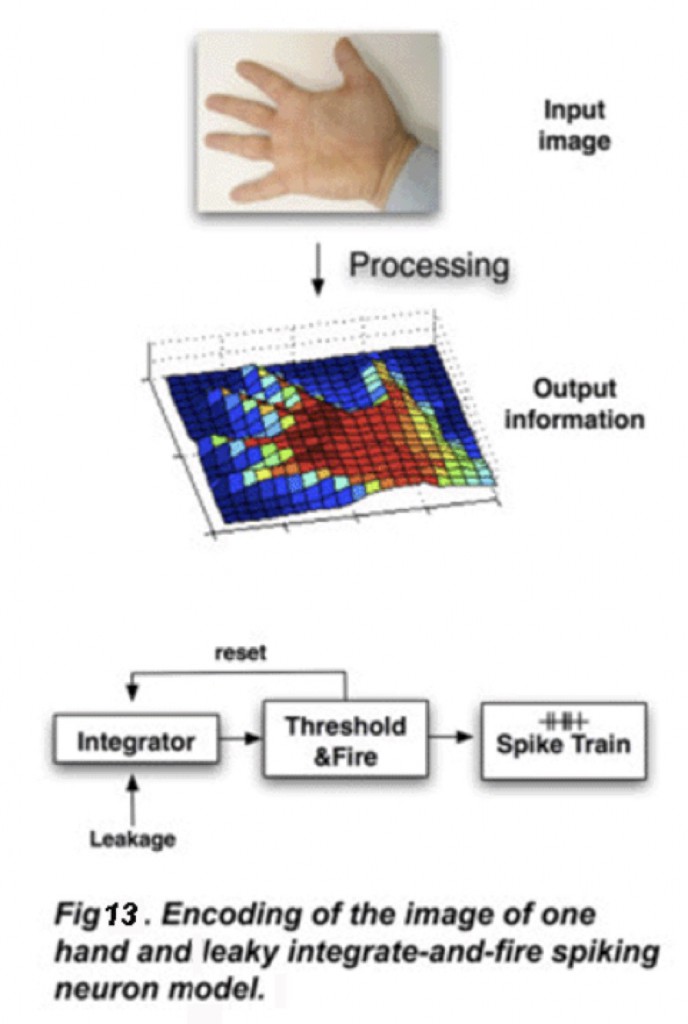
The input images are captured by a photosensor array (preferably a logarithmic response camera that has a similar response to the human visual system and can reduce saturation in high contrast visual scenes). These visual signals are processed by a set of separate spatial and temporal filters that enhance specific features of the captured visual information. The model can take into account the irregular distribution of photoreceptors within the human retina: a high density of pixels and smaller receptive field sizes in central areas; lower density and bigger receptive fields in peripheral areas. A gain factor can be specified for every individual channel as well as a global gain. The next stage reduces the information to the resolution of the electrode matrix, with the option of defining specific receptive field shapes and sizes. Finally, a mapping and neuromorphic coding (into output charge-balanced pulses that can be sent to each electrode) is carried out and feeds the radio frequency link that goes to the microelectrode array. Thus the continuously varying input video stream is converted into neuromorphic pulse-coded signals through a circuit that emulates the function of retinal neurons (Fig. 13) (Fernandez, Pelayo et al. 2005; Morillas, Romero et al. 2007).
The implementation of the model in digital hardware provides a flexible design, achieving a high performance with response times several orders of magnitude lower than that of biological systems. The present version of the whole system is able to work properly up to 40 MHz (Pelayo, Romero et al. 2004; Fernandez, Pelayo et al. 2005; Normann, Greger et al. 2009). This means that 40,000 electrodes could be stimulated with an inter-spike temporal resolution equal to or lower than 1msec. The use of reconfigurable circuitry (FPGA) permits one to adjust or even change the spiking model easily (Pelayo, Romero et al. 2004; Morillas, Romero et al. 2007). Furthermore this technology can be used for psychophysical experimentation in order to determine the best image processing strategy, and to get insights into the optimal number of electrodes, grey levels and grid size (Dagnelie, Keane et al. 2007; Dagnelie 2008).
Finally the output of the bio-inspired retina has to be transmitted to a remote stimulating device that electrically connects with the implanted microelectrode array (Figure 6). Ideally this should be done telemetrically (i.e. without the need of attached wires) to reduce the risk of infections and an opto-coupling stage should be incorporate to protect the patient against electrical risks. Furthermore, for a durable system, the materials used in the implanted system must be biocompatible, and electrical-charge displacements must be within margins of capacitive work, never in an irreversible-faradaic working zone (Cogan, Troyk et al. 2007).
Several factors should be taken into account, especially safety, size and power consumptions. Thus, this stimulating device should be able to operate in real-time for a high number of electrodes, have low power requirements and be flexible enough to generate different waveforms and adapt to different stimuli and image processing conditions. Basically it should be able to solve the problem of transmitting data and power to multiple microelectrodes having, simultaneously, the possibility of remote control and sensing of microelectrode status. It implies a compromise between the power level requirements of the stimulation circuits and the signal bandwidth requirements (Pelayo, Romero et al. 2004; Romero, Morillas et al. 2008).
7. Safe And Effective Stimulation Of Visual Pathways Through Multiple Micro Electrodes.
Any visual neuroprosthetic system must be implanted into the retina, optic nerve, LGN or visual cortex and remain fully functional for periods that may eventually extend to many decades. Therefore these devices must be highly biocompatible and be able to resist the attack of biological fluids, proteases, macrophages or any products of metabolism. Furthermore it is necessary to take into account the possible damage of neural tissues by continual charge injection and the most effective means of stimulating the visual pathways. On the other hand, although there are no major safety concerns stemming from the pilot studies carried out to date, the risks of the surgical procedures involved are not negligible. All these considerations place unique constraints on the architecture, material, and surgical techniques used in the implementation of visual prostheses interfaces (Branner, Stein et al. 2004; Fernandez, Pelayo et al. 2005; Chader, Weiland et al. 2009; Cogan, Ehrlich et al. 2009; Normann, Greger et al. 2009; Shire, Kelly et al. 2009; Marin and Fernandez 2010).
An important problem reported with all available microelectrodes to date, is long-term viability and biocompatibility. Recent studies of the response of neural tissue to stab wound have shown that there are acute and chronic inflammatory reactions which affect both the neural tissue and the surface of the microelectrodes (Hoogerwerf and Wise 1994; Woodford, Carter et al. 1996; Heiduschka and Thanos 1998; Agnew, Duman et al. 1999; Liu, McCreery et al. 1999; Majji, Humayun et al. 1999; Turner, Shain et al. 1999; McCreery, Agnew et al. 2002). These reactions often result in damage to neurons and microelectrodes and lead to the proliferation of a glial scar around the implanted probes which prevents neurons being recorded or stimulated (Polikov, Tresco et al. 2005; Seymour and Kipke 2007; McConnell, Rees et al. 2009). The reasons for the inflammatory response lie in molecular and cellular reactions at foreign surfaces (Edell, Van Toi et al. 1992; Heiduschka and Thanos 1998). These responses can be controlled and one of the big challenges in this field is to create new, more biocompatible surfaces. The solution may involve coating the electrodes with bio-active molecules that are slowly released into the surrounding neural tissue (Marin and Fernandez 2010).
8. Selection Of Suitable Subjects For A Visual Prosthesis.
The selection of a specific person for a visual implant is not straightforward. There are no strict standardized criteria for accepting or rejecting a candidate, nor for the best rehabilitation procedure for every type of blindness. Generally a choice should be made between different approaches and/or rehabilitation procedures depending on availability, efficacy or rejection of invasive methods (Veraart, Duret et al. 2004; Dowling 2005; Dagnelie 2006). But a pre-surgical protocol and improved methods for predicting success with a visual neuroprosthesis need to be developed (Fernandez, Pelayo et al. 2005; Merabet, Rizzo et al. 2007; Dagnelie 2008).
In general, it is considered crucial, at least in these preliminary stages, that the subject should have no residual vision and have had no significant benefit from a conventional visual aid. However, these seemingly straightforward criteria do not always work well in practice and there are different definitions of “residual vision” and “significant benefit from a visual aid”. Needs and wishes of individual subjects are also significant variables for implant candidacy. This issue is further complicated because it is not possible to predict success with a visual implant in a specific person. Clearly, our knowledge regarding visual system anatomy and function may allow for crude bio-inspired models and strategies of stimulation. However, what has not been discussed is how the type, onset, duration and temporal profile of an individual’s visual loss may have repercussions on the success of the device. To decide what time is the most suitable for intervention is a difficult double-faced ethical decision (Veraart, Duret et al. 2004).
Recognizing these limitations, it is generally acknowledged that previous visual experience is necessary for the patient to interpret and recognize the visual patterns that are generated. In this context, it has been presumed that if blindness occurs after the age of ten years of age (i.e. after the critical period), visual pathways should have developed normally and thus remain excitable (Veraart, Duret et al. 2004; Dagnelie 2006).
Improved networking among research groups would clearly help in developing a common body of standardized test and standardized selection criteria (Dagnelie 2008). According to Merabet et al (2007) (Merabet, Rizzo et al. 2007) the criteria for potential users of a visual neuroprosthesis should include:
- Visual function criteria and pre- and post-implantation testing. Indications in favor of a cortical visual implant could include profound and bilateral visual loss, but more objective and quantitative criteria should be developed. These studies need to incorporate a more quantifiable means of estimating risk and benefit for a given candidate.
- Electrophysiological criteria. Measurement of electroretinogram (ERG) as well as visual evoked potentials (VEP) should be a basic component in candidate selection. Improved methods for predicting success with a visual implant need to be developed. These methods could include a test using data from transcranial magnetic stimulation of visual pathways and new imaging techniques such as functional magnetic resonance (fMRI).
- Medical, anatomical and surgical criteria. The ideal candidate should be a healthy adult. The medical history should include the onset and evolution of the blindness, physical examination, and laboratory tests to include or exclude candidates and to assist the implant team in planning a total program, including post-implant training.
- Evaluation of the risks and limitations associated with the surgery and every specific approach. These are usually small for persons in good general health but increase with age and other pathological conditions.
- Quantitative evaluation of the subjects’ needs and performance using standardized psychophysical and behavioral methods.
- Psychological testing to discard psychiatric disorders and other possible mental status that could affect the success of the visual prosthesis.
There are other important issues that should be taken into account and case studies of surgical sight restoration following long-term visual deprivation provide relevant insights (Fine, Wade et al. 2003; Gregory 2003). For example, following ocular surgical procedures aimed at regaining some degree of functional vision, patients blinded for many years experience profound difficulty in various visual tasks, particularly those requiring the identification and recognition of objects. Interestingly, if patients are allowed to explore the same object through touch, they can recognize it immediately as to register their newly acquired visual percepts with their existing senses. These findings demonstrate that even when vision reaches the brain physiologically, visual perception remains nonetheless impaired. Thus, the simple restoration of a lost sensory input may not itself suffice in reconstituting the sense it normally provides. Active visual rehabilitation may be necessary to maximize the adaptation and get the most from these devices. Moreover some form of spatial remapping between the bio-inspired encoder and the stimulation pattern delivered to the array of electrodes implanted in the visual cortex is highly recommended (Normann, Warren et al. 2001; Fernandez, Alfaro et al. 2002; Eckmiller, Neumann et al. 2005; Fernandez, Pelayo et al. 2005; Romero, Morillas et al. 2008). As a result, the right learning, remapping and rehabilitation strategies could potentially help to modulate the plasticity of the brain and contribute to ever-improving performance and more concordant perceptions.
9. Conclusions And Future Perspectives.
The field of visual prosthetics is rapidly emerging and requires extraordinary diverse, lengthy and intimate collaborations among basic scientists, engineers and clinicians. Significant challenges to the development of a successful visual prosthesis include limitations to the number of electrodes, biocompatibility, encapsulation, electrode degradation, power interference, signal and imaging processing, interference with residual vision, functional evaluation and training. Despite all these issues several research groups are developing sophisticated microelectronic devices designed to stimulate viable neuronal tissue in the hopes of generating functional vision artificially.
To date, at least 23 different devices are under development, several groups are pursuing human clinical trials either with acute or chronic implantation of neurostimulating devices and increasing numbers are expected within the next years (Lachman, 2014). Thus this type of technology is clearly expanding and ophthalmology has emerged as one of the more interesting areas of research in the neurostimulation field. However, even the most advanced of these experimental devices has only been able to provide evidence that such an approach is feasible. What remains to be conclusively demonstrated is whether or not the visual percepts produced by implanted microelectronic devices can create meaningful visual perceptions that can be translated into functional gains such as the recognition, localization and grasping of objects or skillful navigation in an unfamiliar environment.
The greater impediments to future progress in visual neuroprosthesis approaches are not only the technical, engineering and surgical issues that remain to be solved, but also the development and implementation of strategies designed to interface with the visually deprived brain specifically tailored for an individual patient’s own needs. This would particularly involve an improved patient selection and a “custom-tailoring” of visual prosthetic devices to the subject. A key issue in this context that has often been utterly underrated is the role of neural plasticity. Thus, these strategies should take into consideration not only standardized methods and employ current clinical and technological expertise, but also consider newly emerging developmental and neurophysiological evidence. For example, there is considerable evidence that adaptive and compensatory changes occur within the brain following the loss of sight (Cohen, Celnik et al. 1997; Pascual-Leone, Hamilton et al. 1999; Bavelier and Neville 2002; Fernandez, Pelayo et al. 2005; Merabet, Rizzo et al. 2005; Ptito and Kupers 2005; Bernabeu, Alfaro et al. 2009). These studies have shown that in some patients the occipital parts of the brain that sighted subjects use to process visual information are transformed and utilized to process tactile and auditory stimuli. This plastic change in the brain probably allows blind subjects to extract greater information from touch and hearing, thus improving quality of life and enhancing the integration of the blind in the social and working environment of a sighted society. The modulation and understanding of these neuroplastic processes is crucial for the success of any visual neuroprosthesis and can therefore provide the neuroscientific foundation for improved rehabilitation and teaching strategies for the blind.
Hopefully the advances in medicine, ophthalmology and genetics will be able to devise new ways of preventing diseases of the retina and visual pathways or in transplanting neurons that have been lost. However genetic science and treatment will not help in injuries due to trauma and probably will not eliminate all the visual impairments due to ageing. Therefore, progress in vision prosthesis technologies is regarded as a necessity for the future. We hope that the progresses in medical technologies, material science and bioengineering, together with the increase of intelligence in these visual neuroprosthetic devices will improve the quality of life of people that are affected by visual impairments, and encourage further development of custom-tailored devices for restoring functional sight in the blind.
References
Agnew, B. J., J. G. Duman, et al. (1999). “Cytological transformations associated with parietal cell stimulation: critical steps in the activation cascade.” J Cell Sci 112(Pt 16): 2639-2646. [PubMed]
Bak, M., J. P. Girvin, et al. (1990). “Visual sensations produced by intracortical microstimulation of the human occipital cortex.” Med Biol Eng Comput 28(257-259). [PubMed]
Balkany, T. J., A. V. Hodges, et al. (2005). “Cochlear implant soft failures consensus development conference statement.” Otol Neurotol 26(4): 815-818. [PubMed]
Barbeau, H., D. A. McCrea, et al. (1999). “Tapping into spinal circuits to restore motor function.” Brain Res Brain Res Rev 30(1): 27-51. [PubMed]
Bavelier, D. and H. J. Neville (2002). “Cross-modal plasticity: where and how?” Nat Rev Neurosci 3(6): 443-452. [PubMed]
Bernabeu, A., A. Alfaro, et al. (2009). “Proton magnetic resonance spectroscopy (1H-MRS) reveals the presence of elevated myo-inositol in the occipital cortex of blind subjects.” Neuroimage 47(4): 1172-1176. [PubMed]
Bialek, W., F. Rieke, et al. (1991). “Reading a neural code.” Science 252(5014): 1854-1857. [PubMed]
Bouccara, D., P. Avan, et al. (2005). “[Auditory rehabilitation].” Med Sci (Paris) 21(2): 190-197. [PubMed]
Bradley, D. C., P. R. Troyk, et al. (2005). “Visuotopic mapping through a multichannel stimulating implant in primate V1.” J Neurophysiol 93(3): 1659-1670. [PubMed]
Branner, A., R. B. Stein, et al. (2004). “Long-term stimulation and recording with a penetrating microelectrode array in cat sciatic nerve.” IEEE Trans Biomed Eng 51(1): 146-157. [PubMed]
Brelen, M. E., V. Vince, et al. (2010). “Measurement of evoked potentials following electrical stimulation of the human optic nerve.” Invest Ophthalmol Vis Sci. [PubMed]
Brindley, G. S. (1982). “Effects of electrical stimulation of the visual cortex.” Hum Neurobiol 1: 281-283. [PubMed]
Brindley, G. S., P. E. K. Donaldson, et al. (1972). “The extent of the region of occipital cortex that when stimulated gives phosphenes fixed in the visual field.” J Physiol (Lond) 225: 57-58. [PubMed]
Brindley, G. S. and W. S. Lewin (1968). “The sensations produced by electrical stimulation of the visual cortex.” J Physiol (Lond) 196: 479-493. [PubMed]
Brors, D. and D. Bodmer (2004). “New aspects of inner ear research.” Hosp Med 65(7): 392-395. [PubMed]
Busskamp, V., S. Picaud, et al. (2012). “Optogenetic therapy for retinitis pigmentosa.” Gene Ther 19(2): 169-175. [PubMed]
Campbell, P. K., K. E. Jones, R. J. Huber, K. W. Horch and R. A. Normann (1991). “A silicon-based, three-dimensional neural interface: manufacturing processes for an intracortical electrode array.” IEEE Trans. Biomed. Eng. 38(8): 758-768. [PubMed]
Carmena, J. M., M. A. Lebedev, et al. (2005). “Stable ensemble performance with single-neuron variability during reaching movements in primates.” J Neurosci 25(46): 10712-10716. [PubMed]
Cha, K., K. W. Horch and R. A. Normann (1992). “Mobility performance with a pixelized vision system.” Vision Res. 32(7): 1367-1372. [PubMed]
Chader, G. J., J. Weiland, et al. (2009). “Artificial vision: needs, functioning, and testing of a retinal electronic prosthesis.” Prog Brain Res 175: 317-332. [PubMed]
Chang, P. (2005). “Implantable hearing devices–beyond hearing aids.” Aust Fam Physician 34(3): 157-161. [PubMed]
Chatelin, V., E. J. Kim, et al. (2004). “Cochlear implant outcomes in the elderly.” Otol Neurotol 25(3): 298-301. [PubMed]
Chow, A. Y. and N. Peachey (1999). “The subretinal microphotodiode array retinal prosthesis II.” Ophthalmic Res 31(3): 246. [PubMed]
Cogan, S. F., J. Ehrlich, et al. (2009). “Sputtered iridium oxide films for neural stimulation electrodes.” J Biomed Mater Res B Appl Biomater 89B(2): 353-361. [PubMed]
Cogan, S. F., P. R. Troyk, et al. (2007). “The influence of electrolyte composition on the in vitro charge-injection limits of activated iridium oxide (AIROF) stimulation electrodes.” J Neural Eng 4(2): 79-86. [PubMed]
Cohen, L. G., P. Celnik, et al. (1997). “Functional relevance of cross-modal plasticity in blind humans.” Nature 389(6647): 180-183. [PubMed]
Cohen, N. L. (2004). “Cochlear implant candidacy and surgical considerations.” Audiol Neurootol 9(4): 197-202. [PubMed]
Dagnelie, G. (2006). “Visual prosthetics 2006: assessment and expectations.” Expert Rev Med Devices 3(3): 315-325. [PubMed]
Dagnelie, G. (2008). “Psychophysical Evaluation for Visual Prosthesis.” Annu Rev Biomed Eng 10: 339-368. [PubMed]
Dagnelie, G., P. Keane, et al. (2007). “Real and virtual mobility performance in simulated prosthetic vision.” J Neural Eng 4(1): S92-101. [PubMed]
de Ruyter van Steveninck, R. R., G. D. Lewen, et al. (1997). “Reproducibility and variability in neural spike trains.” Science 275(5307): 1805-1808. [PubMed]
Degenaar, P., N. Grossman, et al. (2009). “Optobionic vision–a new genetically enhanced light on retinal prosthesis.” J Neural Eng 6(3): 035007. [PubMed]
Delbeke, J., D. Pins, et al. (2001). “Electrical stimulation of anterior visual pathways in retinitis pigmentosa.” Invest Ophthalmol Vis Sci 42(1): 291-297. [PubMed]
Dobelle, W. H. (2000). “Artificial vision for the blind by connecting a television camera to the visual cortex.” Asaio J 46(1): 3-9. [PubMed]
Dobelle, W. H. and M. G. Mladejovsky (1974). “Phosphenes produced by electrical stimulation of human occipital cortex, and their application to the development of a prosthesis for the blind.” J Physiol (Lond) 243: 553-576. [PubMed]
Dobelle, W. H., M. G. Mladejovsky, et al. (1976). “‘Braille’ reading by a blind volunteer by visual cortex stimulation.” Nature 259: 111-112. [PubMed]
Dobelle, W. H., M. G. Mladejovsky, et al. (1974). “Artificial vision for the blind: electrical stimulation of visual cortex offers hope for a functional prosthesis.” Science 183: 440-444. [PubMed]
Donoghue, J. P. (2002). “Connecting cortex to machines: recent advances in brain interfaces.” Nat Neurosci 5 Suppl: 1085-1088. [PubMed]
Donoghue, J. P., A. Nurmikko, et al. (2004). “Development of neuromotor prostheses for humans.” Suppl Clin Neurophysiol 57: 592-606. [PubMed]
Dorn, J. D., A. K. Ahuja, et al. (2013). “The Detection of Motion by Blind Subjects With the Epiretinal 60-Electrode (Argus II) Retinal Prosthesis.” JAMA Ophthalmol 131(2): 183-189. [PubMed]
Dougherty, R. F., V. M. Koch, et al. (2003). “Visual field representations and locations of visual areas V1/2/3 in human visual cortex.” J Vis 3(10): 586-598. [PubMed]
Dowling, J. (2005). “Artificial human vision.” Expert Rev Med Devices 2(1): 73-85. [PubMed]
Dowling, J. (2009). “Current and future prospects for optoelectronic retinal prostheses.” Eye (Lond) 23(10): 1999-2005. [PubMed]
Eckmiller, R. (1997). “Learning retina implants with epiretinal contacts.” Ophthalmic Res 29(5): 281-289. [PubMed]
Eckmiller, R., D. Neumann, et al. (2005). “Tunable retina encoders for retina implants: why and how.” J Neural Eng 2(1): S91-S104. [PubMed]
Edell, D. J., V. Van Toi, et al. (1992). “Factors influencing the biocompatibility of insertable silicon microshafts in cerebral cortex.” IEEE Trans Biomed Eng 39: 635-643. [PubMed]
Fernandez, E., A. Alfaro, et al. (2002). “Mapping of the human visual cortex using image-guided transcranial magnetic stimulation.” Brain Res Protoc. 10: 115-124. [PubMed]
Fernandez, E., J. Ferrandez, et al. (2000). “Population coding in spike trains of simultaneously recorded retinal ganglion cells.” Brain Res 887(1): 222-229. [PubMed]
Fernandez, E., B. Greger, et al. (2014). “Acute human brain responses to intracortical microelectrode arrays: challenges and future prospects.” Front Neuroeng 7: 24. [PubMed]
Fernandez, E., F. Pelayo, et al. (2005). “Development of a cortical visual neuroprosthesis for the blind: the relevance of neuroplasticity.” J Neural Eng 2(4): R1-R12. [PubMed]
Fine, I., A. R. Wade, et al. (2003). “Long-term deprivation affects visual perception and cortex.” Nat Neurosci 6(9): 915-916. [PubMed]
Galvani, L. and G. Aldini, De Viribus Electricitatis In Motu Musculari Comentarius Cum Joannis Aldini Dissertatione Et Notis; Accesserunt Epistolae ad animalis electricitatis theoriam pertinentes. 1792: Apud Societatem Typographicam.
Gregory, R. L. (2003). “Seeing after blindness.” Nat Neurosci 6(9): 909-910. [PubMed]
Greschner, M., M. Bongard, et al. (2002). “Retinal ganglion cell synchronization by fixational eye movements improves feature estimation.” Nat Neurosci 5(4): 341-347. [PubMed]
Grill-Spector, K. and R. Malach (2004). “The human visual cortex.” Annu Rev Neurosci 27: 649-677. [PubMed]
Gross, C. G. (1997). From Imhotep to Hubel and Wiesel: the story of visual cortex. New York, Plenum Press. [Direct pdf link]
Heiduschka, P. and S. Thanos (1998). “Implantable bioelectric interfaces for lost nerve functions.” Prog. Neurobiol. 55: 433-461. [PubMed]
Hochberg, L. R., M. D. Serruya, et al. (2006). “Neuronal ensemble control of prosthetic devices by a human with tetraplegia.” Nature 442(7099): 164-171. [PubMed]
Hoogerwerf, A. C. and K. D. Wise (1994). “A 3D micro-electrode array for chronic neural recording.” IEEE Trans Biomed Eng 41: 1136-1146. [PubMed]
Hubel, D. H. and T. N. Wiesel (1977). “Functional architecture of macaque monkey visual cortex (Ferrier Lecture).” Proc R Soc Lond B 198: 1-59. [PubMed]
Hubel, D. H. and T. N. Wiesel (1990). Brain mechanism of vision. The Perceptual World. I. Rock. New York, W.H. Freeman: 3-24.
Humayun, M. S., J. D. Dorn, et al. (2012). “Interim results from the international trial of Second Sight’s visual prosthesis.” Ophthalmology 119(4): 779-788. [PubMed]
Humayun, M. S., J. Weiland, et al. (2003). “Visual perception in a blind subject with a chronic microelectronic retinal prosthesis.” Vision Res 43: 2573-2581. [PubMed]
Hunter, T. B., M. T. Yoshino, et al. (2004). “Medical devices of the head, neck, and spine.” Radiographics 24(1): 257-285. [PubMed]
Jeffries, A. M., N. J. Killian, et al. (2014). “Mapping the primate lateral geniculate nucleus: a review of experiments and methods.” J Physiol Paris 108(1): 3-10. [PubMed]
Jones, B. W., M. Kondo, et al. (2012). “Retinal remodeling.” Jpn J Ophthalmol 56(4): 289-306. [PubMed]
Jones, B. W. and R. E. Marc (2005). “Retinal remodeling during retinal degeneration.” Exp Eye Res 81(2): 123-137. [PubMed]
Jones, B. W., C. B. Watt, et al. (2003). “Retinal remodeling triggered by photoreceptor degenerations.” J Comp Neurol 464(1): 1-16. [PubMed]
Kane, S. R., S. F. Cogan, et al. (2013). “Electrical performance of penetrating microelectrodes chronically implanted in cat cortex.” IEEE Trans Biomed Eng 60(8): 2153-2160. [PubMed]
Kitiratschky, V. B., K. Stingl, et al. (2015). “Safety evaluation of “retina implant alpha IMS”–a prospective clinical trial.” Graefes Arch Clin Exp Ophthalmol 253(3): 381-387. [PubMed]
Kolb, H., et al. (2005). Webvision: The Neural Organization of Retina and Visual System (available at: http://webvision.med.utah.edu).
Kolb, H., R. Nelson, et al. (2001). “Cellular organization of the vertebrate retina.” Prog Brain Res 131: 3-26. [PubMed]
Lewis, P. M., H. M. Ackland, et al. (2015). “Restoration of vision in blind individuals using bionic devices: a review with a focus on cortical visual prostheses.” Brain Res 1595: 51-73. [PubMed]
Liu, X., D. B. McCreery, et al. (1999). “Stability of the interface between neural tissue and chronically implanted intracortical microelectrodes.” IEEE Trans Rehabil Eng 7(3): 315-326. [PubMed]
Lachman M. (2014). “Eye disorders are focus of novel neurostimulation technologies”. Medtech insight. February 2014.
Loeb, G. E. (1990). “Cochlear prosthetics.” Annu Rev Neurosci 13: 357-371. [PubMed]
Majji, A. B., M. S. Humayun, et al. (1999). “Long-term histological and electrophysiological results of an inactive epiretinal electrode array implantation in dogs.” Invest Ophthalmol Vis Sci 40(9): 2073-2081. [PubMed]
Marc, R., R. Pfeiffer, et al. (2014). “Retinal prosthetics, optogenetics, and chemical photoswitches.” ACS Chem Neurosci 5(10): 895-901. [PubMed]
Marc, R. E. and B. W. Jones (2003). “Retinal remodeling in inherited photoreceptor degenerations.” Mol Neurobiol 28(2): 139-147. [PubMed]
Marin, C. and E. Fernandez (2010). “Biocompatibility of intracortical microelectrodes: current status and future prospects.” Front Neuroengineering 3: 8. [PubMed]
Maynard, E. M. (2001). “Visual prostheses.” Annu Rev Biomed Eng 3: 145-168. [PubMed]
McConnell, G. C., H. D. Rees, et al. (2009). “Implanted neural electrodes cause chronic, local inflammation that is correlated with local neurodegeneration.” J Neural Eng 6(5): 56003. [PubMed]
McCreery, D., W. F. Agnew, et al. (2002). “The effects of prolonged intracortical microstimulation on the excitability of pyramidal tract neurons in the cat.” Ann Biomed Eng 30: 107-119. [PubMed]
McDermott, H. J. (2004). “Music perception with cochlear implants: a review.” Trends Amplif 8(2): 49-82. [PubMed]
Merabet, L. B., J. F. Rizzo, 3rd, et al. (2007). “‘Who is the ideal candidate?’: decisions and issues relating to visual neuroprosthesis development, patient testing and neuroplasticity.” J Neural Eng 4(1): S130-135. [PubMed]
Merabet, L. B., J. F. Rizzo, et al. (2005). “What blindness can tell us about seeing again: merging neuroplasticity and neuroprostheses.” Nat Rev Neurosci 6(1): 71-77. [PubMed]
Morillas, C. A., S. F. Romero, et al. (2007). “A neuroengineering suite of computational tools for visual prostheses.” Neurocomputing 70: 2817-2827. [ScienceDirect]
Morillas, C. A., S. F. Romero, et al. (2007). “A design framework to model retinas.” Biosystems 87(2-3): 156-163. [PubMed]
Nakauchi, K., T. Fujikado, et al. (2007). “Threshold suprachoroidal-transretinal stimulation current resulting in retinal damage in rabbits.” J Neural Eng 4(1): S50-57. [PubMed]
Nayagam, D. A., R. A. Williams, et al. (2014). “Chronic electrical stimulation with a suprachoroidal retinal prosthesis: a preclinical safety and efficacy study.” PLoS ONE 9(5): e97182. [PubMed]
Nicolelis, M. A. (2003). “Brain-machine interfaces to restore motor function and probe neural circuits.” Nat Rev Neurosci 4(5): 417-422. [PubMed]
Nicolelis, M. A. and J. K. Chapin (2002). “Controlling robots with the mind.” Sci Am 287(4): 46-53. [PubMed]
Nishida, K., M. Kamei, et al. (2010). “Efficacy of suprachoroidal-transretinal stimulation in a rabbit model of retinal degeneration.” Invest Ophthalmol Vis Sci 51(4): 2263-2268. [PubMed]
Normann, R. A. (2007). “Technology insight: future neuroprosthetic therapies for disorders of the nervous system.” Nat Clin Pract Neurol 3(8): 444-452. [PubMed]
Normann, R. A., B. A. Greger, et al. (2009). “Toward the development of a cortically based visual neuroprosthesis.” J Neural Eng 6(3): 035001. [PubMed]
Normann, R. A., E. Maynard, et al. (1996). “Cortical implants for the blind.” IEEE Spectrum(May 1996): 54-59. [IEEE]
Normann, R. A., D. J. Warren, et al. (2001). “High-resolution spatio-temporal mapping of visual pathways using multi-electrode arrays.” Vision Res 41(10-11): 1261-1275. [PubMed]
Obeid, I., C. Veraart, et al. (2010). “Estimation of phosphene spatial variability for visual prosthesis applications.” Artif Organs 34(5): 358-365. [PubMed]
Paninski, L., M. R. Fellows, et al. (2004). “Spatiotemporal tuning of motor cortical neurons for hand position and velocity.” J Neurophysiol 91(1): 515-532. [PubMed]
Pascual-Leone, A., R. Hamilton, et al. (1999). Neuroplasticity in the adjustment to blindness. Neuronal Plasticity: Building a Bridge from the laboratory to the Clinic. J.Grafman and Y. Christen. Berlin Heidelber New York, Springer-Verlag. [Springer]
Patil, P. G., J. M. Carmena, et al. (2004). “Ensemble recordings of human subcortical neurons as a source of motor control signals for a brain-machine interface.” Neurosurgery 55(1): 27-35; discussion 35-28. [PubMed]
Pelayo, F. J., S. Romero, et al. (2004). “Translating image sequences into spike patterns for cortical neuro-stimulation.” Neurocomputing 58-60: 885-892. [ScienceDirect]
Pena, C., K. Bowsher, et al. (2004). “FDA-approved neurologic devices intended for use in infants, children, and adolescents.” Neurology 63(7): 1163-1167. [PubMed]
Penfield, W. and H. Jaspers (1974). Epilepsy and the functional anatomy of the human brain. London, England, Churchill. [Oxford Press]
Penfield, W. and T. Rasmussen (1950). The cerebral cortex of man. New York, Macmillan. [Wiley]
Pezaris, J. S. and E. N. Eskandar (2009). “Getting signals into the brain: visual prosthetics through thalamic microstimulation.” Neurosurg Focus 27(1): E6. [PubMed]
Pezaris, J. S. and R. C. Reid (2007). “Demonstration of artificial visual percepts generated through thalamic microstimulation.” Proc Natl Acad Sci U S A 104(18): 7670-7675. [PubMed]
Polikov, V. S., P. A. Tresco, et al. (2005). “Response of brain tissue to chronically implanted neural electrodes.” J Neurosci Methods 148(1): 1-18. [PubMed]
Pollen, D. A. (1975). Some perceptual effects of electrical stimulation of the visual cortex in man. New York: Raven.
Ptito, M. and R. Kupers (2005). “Cross-modal plasticity in early blindness.” J Integr Neurosci 4(4): 479-488. [PubMed]
Rizzo, J. F., 3rd, J. Wyatt, et al. (2001). “Retinal prosthesis: an encouraging first decade with major challenges ahead.” Ophthalmology 108(1): 13-14. [PubMed]
Rizzo, J. F., 3rd, J. Wyatt, et al. (2003). “Perceptual efficacy of electrical stimulation of human retina with a microelectrode array during short-term surgical trials.” Invest Ophthalmol Vis Sci 44(12): 5362-5369. [PubMed]
Romero, S. F., C. A. Morillas, et al. (2008). “Computer-controlled neurostimulation for a visual implant.” Biodevices 1: 84-91.
Roska, B., V. Busskamp, et al. (2013). “[Retinitis pigmentosa: eye sight restoration by optogenetic therapy].” Biol Aujourdhui 207(2): 109-121. [PubMed]
Sahel, J. A. and B. Roska (2013). “Gene therapy for blindness.” Annu Rev Neurosci 36: 467-488. [PubMed]
Sanchez, J. C., J. M. Carmena, et al. (2004). “Ascertaining the importance of neurons to develop better brain-machine interfaces.” IEEE Trans Biomed Eng 51(6): 943-953. [PubMed]
Santos, A., M. S. Humayun, et al. (1997). “Preservation of the inner retina in retinitis pigmentosa. A morphometric analysis.” Arch Ophthalmol 115(4): 511-515. [PubMed]
Schmid, E. W., W. Fink, et al. (2014). “Operational challenges of retinal prostheses.” Med Eng Phys 36(12): 1644-1655. [PubMed]
Schmidt, E. M., M. J. Bak, et al. (1996). “Feasibility of a visual prosthesis for the blind based on intracortical microstimulation of the visual cortex.” Brain 119(Pt 2): 507-522. [PubMed]
Schmolesky, M. (2005). Primary Visual Cortex. Webvision: The Neural Organization of Retina and Visual System (available at: http://webvision.med.utah.edu/book/part-ix-psychophysics-of-vision/the-primary-visual-cortex/). H. Kolb, E. Fernandez and R. Nelson.
Schnitzer, M. J. and M. Meister (2003). “Multineuronal firing patterns in the signal from eye to brain.” Neuron 37(3): 499-511. [PubMed]
Serruya, M. D., N. G. Hatsopoulos, et al. (2002). “Instant neural control of a movement signal.” Nature 416: 141-142. [PubMed]
Seymour, J. P. and D. R. Kipke (2007). “Neural probe design for reduced tissue encapsulation in CNS.” Biomaterials 28(25): 3594-3607. [PubMed]
Sherman, S. and R. Guillery (2001). Exploring the thalamus. San Diego, CA, Academic Press.
Shire, D. B., S. K. Kelly, et al. (2009). “Development and implantation of a minimally invasive wireless subretinal neurostimulator.” IEEE Trans Biomed Eng 56(10): 2502-2511. [PubMed]
Shivdasani, M. N., N. C. Sinclair, et al. (2014). “Factors affecting perceptual thresholds in a suprachoroidal retinal prosthesis.” Invest Ophthalmol Vis Sci 55(10): 6467-6481. [PubMed]
Smirnakis, S. M., M. J. Berry, et al. (1997). “Adaptation of retinal processing to image contrast and spatial scale.” Nature 386(6620): 69-73. [PubMed]
Stieglitz, T., M. Schuettler, et al. (2004). “Neural prostheses in clinical applications–trends from precision mechanics towards biomedical microsystems in neurological rehabilitation.” Biomed Tech (Berl) 49(4): 72-77. [PubMed]
Stingl, K., K. U. Bartz-Schmidt, et al. (2015). “Subretinal Visual Implant Alpha IMS – Clinical trial interim report.” Vision Res 111(Pt B): 149-160. [PubMed]
Stone, J. L., W. E. Barlow, et al. (1992). “Morphometric analysis of macular photoreceptors and ganglion cells in retinas with retinitis pigmentosa.” Arch Ophthalmol 110(11): 1634-1639. [PubMed]
Stronks, H. C. and G. Dagnelie (2014). “The functional performance of the Argus II retinal prosthesis.” Expert Rev Med Devices 11(1): 23-30. [PubMed]
Tassicker, G. E. (1956). Retinal Stimulator. No. 2, 760, 483. [PubMed]
Trenholm, S. and B. Roska (2014). “Cell-type-specific electric stimulation for vision restoration.” Neuron 83(1): 1-2. [PubMed]
Turner, J. N., W. Shain, et al. (1999). “Cerebral astrocyte response to micromachined silicon implants.” Exp Neurol 156: 33-49. [PubMed]
Veraart, C., F. Duret, et al. (2004). “Vision rehabilitation in the case of blindness.” Expert Rev Med Devices 1(1): 139-153. [PubMed]
Veraart, C., M. C. Wanet-Defalque, et al. (2003). “Pattern recognition with the optic nerve visual prosthesis.” Artif Organs 27(11): 996-1004. [PubMed]
Walter, P., P. Szurman, et al. (1999). “Successful long-term implantation of electrically inactive epiretinal microelectrode arrays in rabbits.” Retina 19(6): 546-552. [PubMed]
Wandell, B. A. and A. R. Wade (2003). “Functional imaging of the visual pathways.” Neurol Clin 21(2): 417-443, vi. [PubMed]
Warren, D. J., E. Fernandez, et al. (2001). “High-resolution two-dimensional spatial mapping of cat striate cortex using a 100-microelectrode array.” Neuroscience 105(1): 19-31. [PubMed]
Wickelgren, I. (2006). “Biomedical engineering. A vision for the blind.” Science 312(5777): 1124-1126. [PubMed]
Wilke, S. D., A. Thiel, et al. (2001). “Population coding of motion patterns in the early visual system.” J Comp Physiol [A] 187(7): 549-558. [PubMed]
Woodford, B. J., R. R. Carter, et al. (1996). “Histopathologic and physiologic effects of chronic implantation of microelectrodes in sacral spinal cord of the cat.” J Neuropathol Exp Neurol 55(9): 982-991. [PubMed]
Yan, Y., X. Sui, et al. (2015). “Spatial characteristics of evoked potentials elicited by a MEMS microelectrode array for suprachoroidal-transretinal stimulation in a rabbit.” Graefes Arch Clin Exp Ophthalmic. [PubMed]
Zrenner, E. (2002). “Will retinal implants restore vision?” Science 295(5557): 1022-1025. [PubMed]
Zrenner, E. (2013). “Fighting blindness with microelectronics.” Sci Transl Med 5(210): 210ps216. [PubMed]
About the Authors
Dr. Eduardo Fernandez was born in Elda (Spain) in 1962. He received an M.D. degree from the University of Alicante (Spain) in 1986 and a Ph. D. degree in Neurosciences with honors in 1990. His collaboration with Dr. Helga Kolb and Dr. Richard Normann, began with a stay from 1990 to 1991 under a postdoctoral fellowship of the Spanish Government (FPI program). He is currently Director of the Department of Histology and Anatomy of the University Miguel Hernández (Spain) and Director of the Biomedical Neuroengineering Research Unit of the Biomedical Research Networking Center on Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN, Spain). His main interest is to develop innovative interfaces with the human nervous system and to develop a two-way direct communication with neurons and ensembles of neurons, which could enhance the life of the disabled. He is also working on the development of new therapeutic approaches for retinal degenerative diseases in order to preserve eyesight and slow disease progression. In recent years he has been working in close collaboration with Dr. Normann on the development of a cortical visual neuroprosthesis that will be able to provide a limited but useful sense of vision to profoundly blind people. Contact Eduardo at e.fernandez@umh.es
Richard A. Normann received the BS, MS and Ph.D. degrees from the University of California, Berkeley, CA,
USA. His original interests were in retinal physiology elucidating photoreceptor adaptation and horizontal cell function. After a 6-year stint in the Neurophysiology lab at the NINDS, National Institutes of Health, he moved to the University of Utah, Department of Bioengineering. His research interests changed from intracellular recordings in retina to a neuroengineering bent. Dr. Normann invented the Utah Electrode Array originally for use as a cortical neuroprosthesis for the blind. Since then his research has spearheaded the use of the UEA in sensory and motor physiology, and as an interface to the central and peripheral nervous systems. He is presently a Distinguished Professor (emeritus) in the Departments of Bioengineering and Ophthalmology, University of Utah. Contact Dr. Normann at normann@utah.edu