The Science Behind Myopia
Brittany J. Carr and William K. Stell
INTRODUCTION
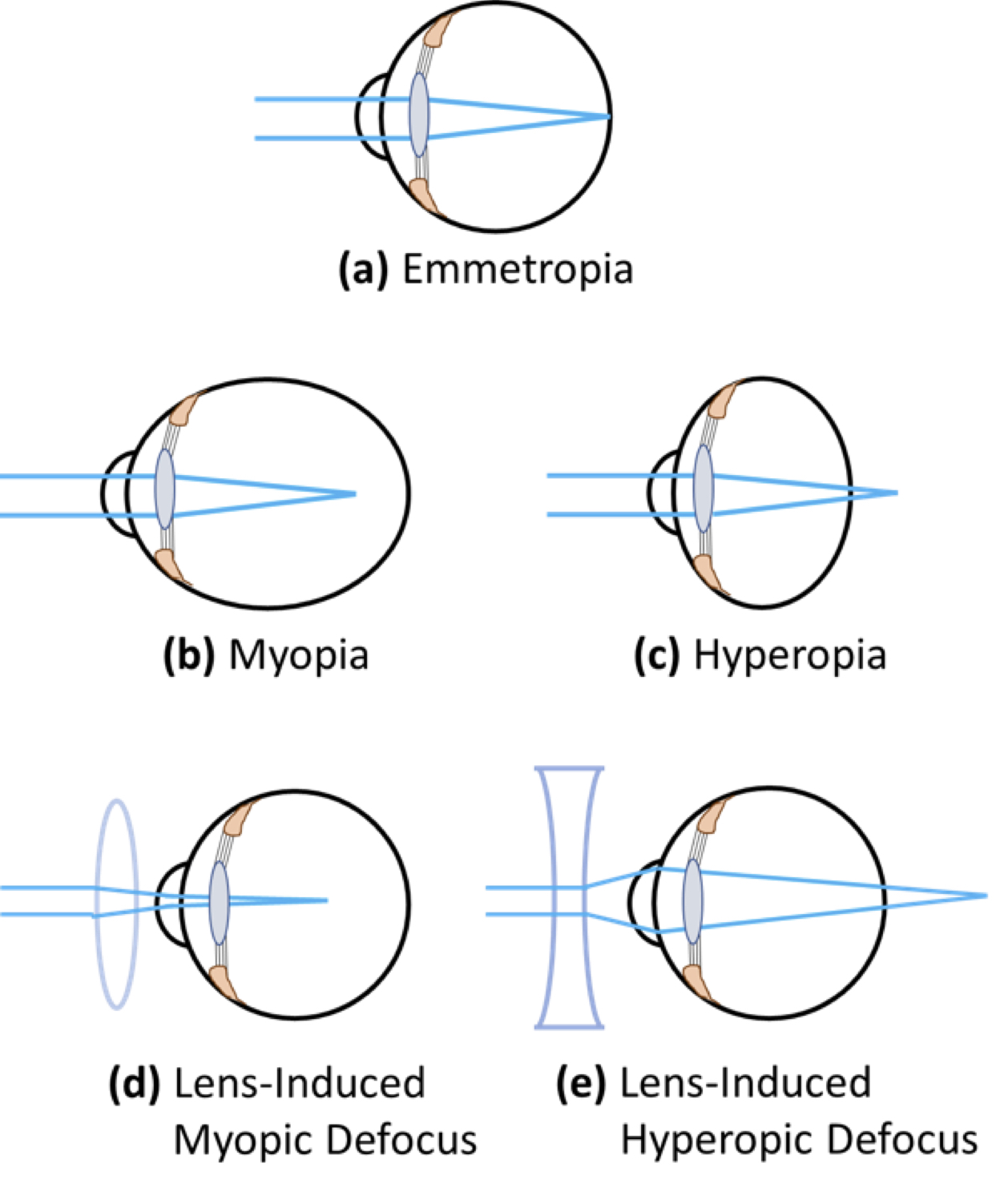
Myopia (near-sightedness) is the most common refractive vision disorder in children. It is characterized by blurring of objects viewed at a distance, and is commonly the result of abnormal elongation of the eyeball – which causes the refractive image formed by the cornea and the lens to fall in front of the photoreceptors of the retina (Fig. 1).
Figure 1. Exaggerated representation of simple refractive errors caused by abnormal eye growth. Emmetropia (normal vision) is the state of refraction in which light is focussed perfectly on retinal rod and cone photoreceptors (a). Myopia (near- or short-sightedness) occurs when the axial length of the eye is too long, and light is focussed in front of the photoreceptors (b). Hyperopia (far-sightedness) occurs when the axial length of the eye is too short, and light is focussed behind the photoreceptors (c). Refractive error can be modified using lenses: In an emmetropic eye, positive lenses impose myopic defocus (d), and negative lenses impose hyperopic defocus (e).
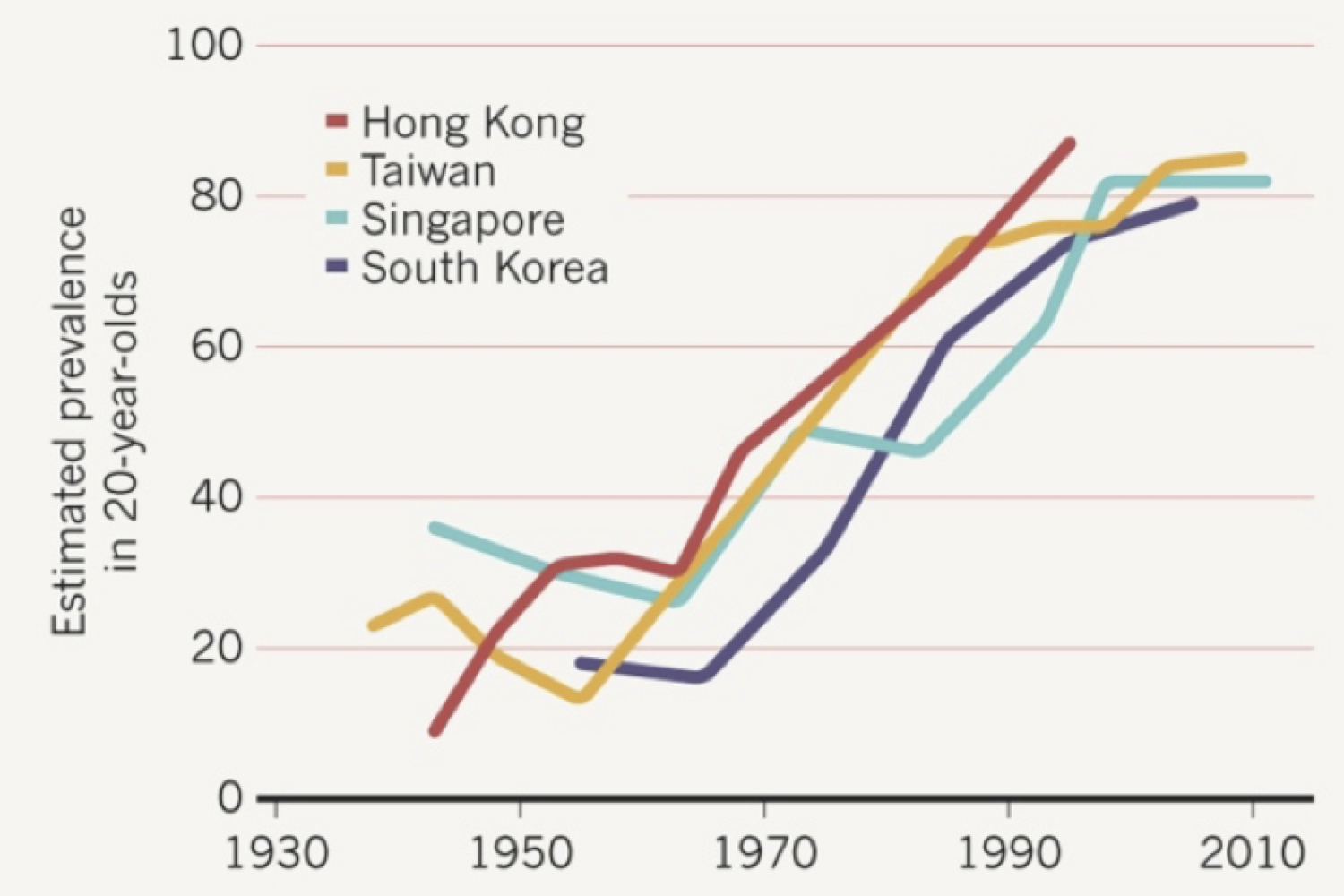
The underlying biological cause of myopia is unknown, and there is no widely accepted means of prevention or cure. The optical error of myopia can be corrected only by using spectacle or contact lenses or corneal surgery. If left untreated, moderate myopia is one of the leading causes of visual impairment worldwide. The greater the degree of myopia, the greater the risk of complications such as macular degeneration, retinal detachment, cataracts, and glaucoma [1]; the risk is especially great when the negative refractive error is more negative than -6.00 D (diopters), a condition called ‘high myopia’. The prevalence of myopia varies greatly, depending on ethnicity, geographical location, and socioeconomic status, but is rising rapidly in most populations studied [2] (Fig. 2). Myopia is a centuries-old problem, and although we have made great progress in scientific investigation of its underlying cause, we have been unsuccessful in preventing it from becoming named by the World Health Organization as an ever-increasing global health concern [3].
Figure 2. Myopia prevalence has been increasing steadily since the 1950’s, especially in Southeast Asian countries. Reproduced with permission from Dolgin, E. 2015 [2]. Figure source: Ian Morgan, Australian National University.
Clinical Aspects & Health Care Burden
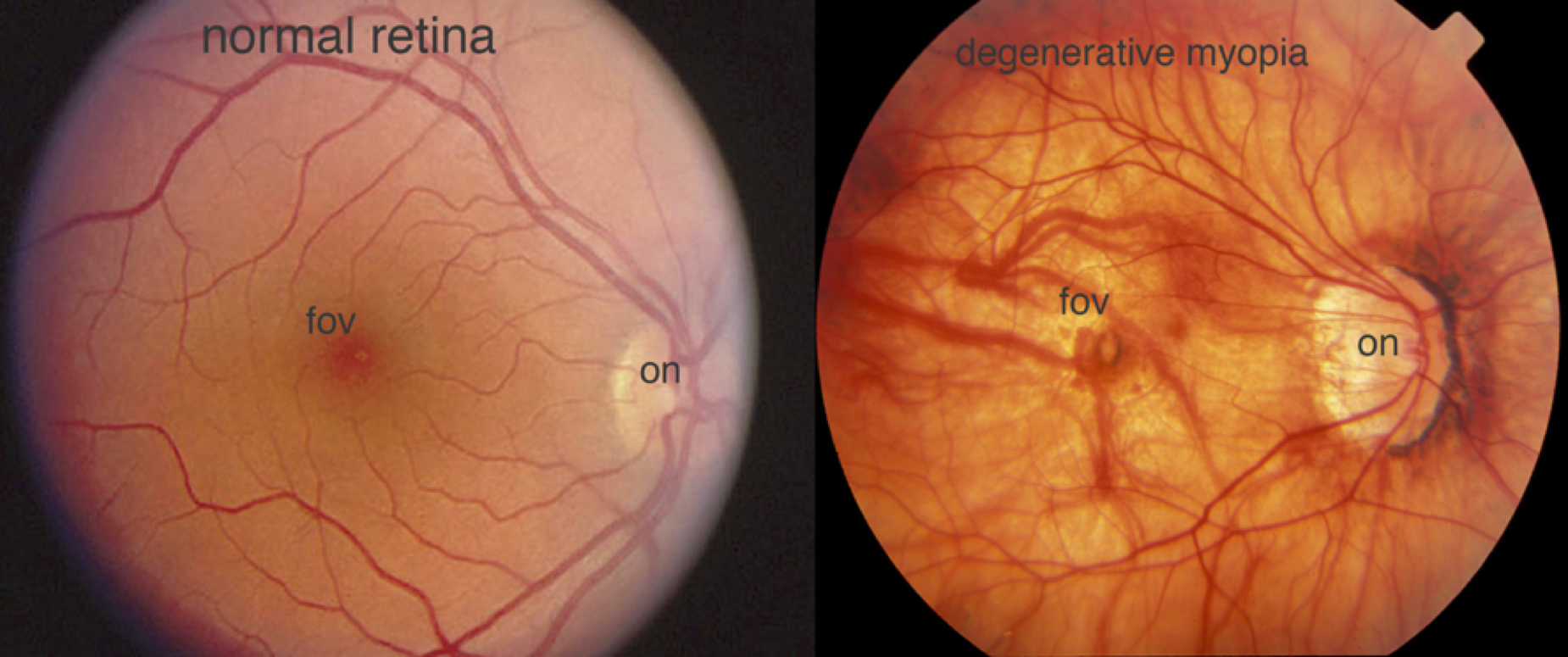
There are two clinical designations for myopia: pathological degenerative myopia, and spontaneous-onset or school age myopia. Pathological myopia is characterized by rapid, extreme axial elongation of the globe, leading to a high refractive error (typically far more negative than -6.00D). This extreme stretching puts stress on the ocular structures (retina, choroid, and sclera), which can then result in degenerative changes in the eye (Fig. 3), and irreversible vision loss. Fortunately, pathological myopia is relatively rare, affecting 0.9-3.1% of the population [4].
Figure 3. A fundus photograph of the retina of a person with degenerative myopia (right) is compared with that of a normal person’s retina (left). The myope’s retina is stretched and thinned, with thin blood vessels, a distorted optic nerve head (on), some folds in the thin retina, and pigmentation at the fovea (fov). Fundus photos provided by James Gilman and the Moran Eye Center ophthalmic photography department.
The most common form of myopia is school-age myopia; this is the form that will be discussed in this chapter, and it will be called simply “myopia” from this point on. It progresses slowly, and usually stabilizes by the age of 20. The retina looks normal. Recently, is has been projected that 2.5 billion people (1/3 of the world’s population) will have some degree of myopia by 2030 [2].
GENES MAY PLAY A ROLE IN DEVELOPMENT OF MYOPIA
Myopia prevalence varies greatly in different ethnic and geographical populations [1]. Recent studies of American preschool children (aged 6–72 months) revealed a prevalence of 1.2% in non-Hispanic whites, 3.7% in Hispanics, 3.98% in Asians, and 6.6% in African Americans [5-6]. Among older children, the difference between Asian and Caucasian populations is obvious; a study of Australian adolescents reported prevalence of 42.7% in 12 yr old and 59.1% in 17 yr old children of East Asian ethnicity, compared to 8.3% in 12 yr old and 17.7% in 17 yr old children of European Caucasian descent [7].
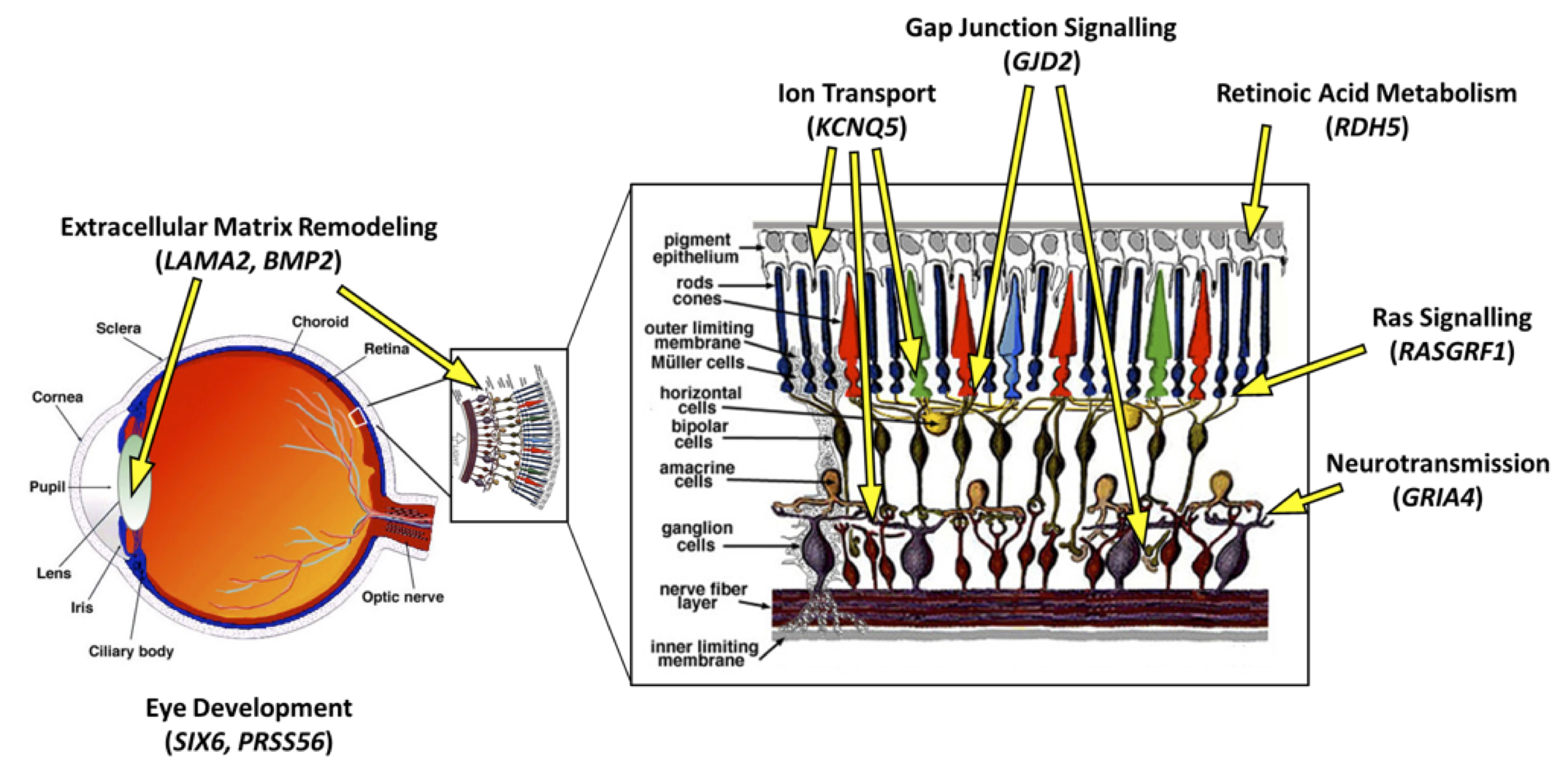
There have been attempts to identify genes that may be important in myopia development. “Genome-wide association studies” (GWAS), which examine genetic variations between individuals to determine whether certain variants are associated with a specific trait (in this case myopia), have identified correlations between variations in certain genes and an increased risk of myopia. The recently published Consortium for Refractive Error And Myopia (CREAM) study is the largest international genome-wide meta-analysis on myopia and refractive error ever conducted. It is based on data from 32 studies encompassing Europe, the United States, Australia and Asia [8]. Twenty-four genomic variations were associated with up to a 10x increase in risk of myopia. These genetic variations are involved in many apparently unrelated ocular functions, instead of a single underlying system or structure. Thus, the CREAM study provided significant evidence that the risk of developing myopia may be affected by many unrelated genetic abnormalities, instead of a single underlying cause (Fig. 4).
Figure 4. Localization and functions of a few genes identified by the CREAM study [8] in the eye and retinal tissues. Some of the genes identified are functionally involved in processes that facilitate communication between cells in the retina (KCNQ5, GJD2, RASGFR1, GRIA4) or control the ability of photoreceptors to respond to light (RDH5). Some others are involved in pre-natal eye growth and development (LAMA2, BMP2, SIX6, PRSS56).
Twin studies and parental myopia studies have also been conducted to try to assess the role of genetics in development of myopia (review9). Twin studies have reported that the correlation of prevalence of myopia was significantly higher in identical twins than fraternal twins [9-11]. Age and the methods used to gather and analyze the data are important, however, as myopia correlations determined by such studies can range from r = 0.11 (low correlation) to r = 0.94 (high correlation) [9]. There is strong evidence to suggest that increased risk of myopia is highly correlated with the number of myopic parents [12-16], and that this risk is independent of environmental factors [12,13]. One problem with twin and familial studies, however, is that few of them bothered to control for environmental factors that would be likely be shared among family members – for example, the likelihood that identical twins engage in the same activities in a much higher frequency than fraternal twins or non-twin siblings.
It is reasonable to assume that genes play a part in myopia risk, but they alone cannot account for the rapid changes in myopia prevalence that are being observed all over the world, nor for the dramatic differences in myopia prevalence within the same ethnic populations – in urban vs. rural environments, for example [2,17]. Thus, the underlying cause of spontaneous-onset myopia is most likely to be a combination of genes and environmental triggers. Most of the identified risk factors for myopia are correlated with environmental influences, such as an increased socioeconomic status, residing in an urban environment, higher education and IQ, increased time spent performing near work, and increased time spent indoors [1,18]. These risk factors are based on observations of human populations over many decades, but they do not provide a mechanistic explanation as to why they are associated with an increased risk of myopia.
To understand the roles of environmental risk factors for the cause of myopia (which are difficult to study in human subjects under stringently controlled conditions), scientists have turned to animal models. The results have demonstrated clearly that, while genetics may determine myopia susceptibility (e.g.: age of onset, rate of progression, ultimate refractive error), environmental factors are powerful modifiers of eye growth, and they may override genetic predispositions. For a comprehensive review on the biological basis and mechanisms of the eye’s self-regulation of growth see, “Homeostasis of eye growth and the question of myopia”, by Wallman & Winawer [19].
ANIMAL MODELS OF MYOPIA: WHAT WE SEE CONTROLS HOW THE EYE GROWS
The first reported case of myopia in experimental animals was observed serendipitously in non-human primates, during investigations into the cortical effects of monocular visual deprivation in young macaques. In these animals, sewing the eyelids shut for many months resulted in high levels of myopia20; the longer the period of visual deprivation, the greater the resultant refractive error21. The establishment of this “form-deprivation” model of myopia in primates led to parallel studies of experimentally-induced myopia in young tree shrews [22] and chickens [23]. Instead of sewing the eyelids shut, scientists now use frosted plastic goggles to induce deprivation myopia, or lenses with a strong negative power to cause “lens-induced myopia” [24] (Fig. 5). Today, the most commonly-used myopia model animals are chicks and guinea pigs, but tree shrews, non-human primates, and mice are also studied [25].
Figure 5. The effects of goggles on the initially focused image of a square-wave grating. When no goggle is applied, the grating image appears as sharply defined black and white lines (a). Myopia can be induced by manipulating retinal images optically, in animal models such as the chick, by application of a frosted diffuser goggle (b) or a negative-powered lens (c). Both treatments reduce spatial contrast of the image (difference between white and black), especially at higher spatial frequencies (the sharpness of the line borders).
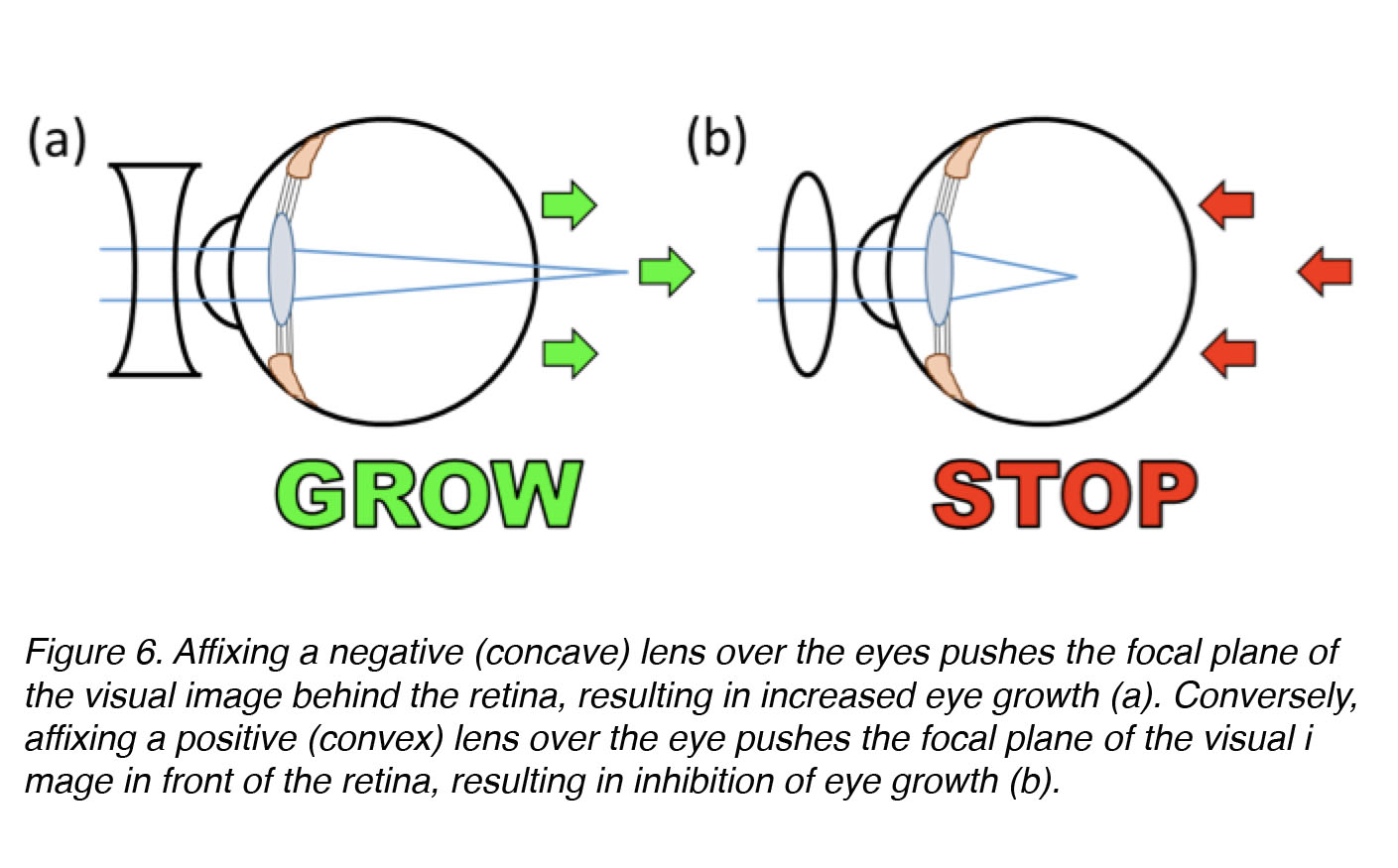
That we can experimentally-induce myopia in young, rapidly growing animals tells us that the visual environment plays an important role in regulation of eye growth. Applying goggles over the eye works even when the optic nerve has been cut, which tells us that the eye alone – or more specifically, a specialized sensorineural tissue in the eye called the retina – can discriminate whether the image is well focussed or not, and act locally to mediate optically-induced changes in eye growth [19,26,27]. Affixing a frosted goggle or negative lens over the eye causes excessive axial elongation of the eye, and myopia, but affixing a positive lens over the eye causes inhibition of axial elongation, and far-sightedness (hyperopia) (Fig. 6). The complementary effects of positive and negative defocus reveal that the eye – most probably the retina itself – can discriminate between positive and negative defocus, as well as between unfocussed blur (form-deprivation) and blur due to negative lens treatment, even though the static images produces by the two different treatments appear very similar (Fig. 5, b vs. c). We do not know whether eye growth is controlled by two different signals, one “start” and one “stop”, operating in push-pull fashion (like turning the steering of an automobile to left or right) – or by a single signal that increases elongation (“grow” or “on”) or decreases it (“stop” or “off”) (like increasing or decreasing pressure on the accelerator pedal of an automobile). We also do not know the identities of these hypothetical “start” or “stop” growth signals, but a number of signalling substances have been identified that are very strong candidates for “eye growth regulators” (discussed in greater detail below).
Figure 6. Affixing a negative (concave or diverging) lens over the eye pushes the focal plane of the visual image behind the retina, inducing an increase in axial elongation and a more myopic refraction (a). Conversely, affixing a positive (convex or converging) lens over the eye pushes the focal plane of the visual image in front of the retina, inducing a decrease in axial elongation and a more hyperopic refraction (b).
Myopia-inducing stimuli require prolonged exposures (days-weeks) for a significant effect. In the chick, however, it takes only ≥2 hrs per day of unobstructed viewing of a richly textured visual environment to completely block the development of form-deprivation myopia [28], and positive-lens wear for only 2 min, four times per day, is enough to block lens-induced myopia [29]. In addition, the eye can “recover” from experimentally induced myopia and return to an emmetropic refractive state, if the animal (or human) is young enough. Once an organism has reached adulthood, however, there is no known way to decrease the size of the globe, without surgical intervention.
INHIBITING ACCOMMODATION DOES NOT PREVENT MYOPIA
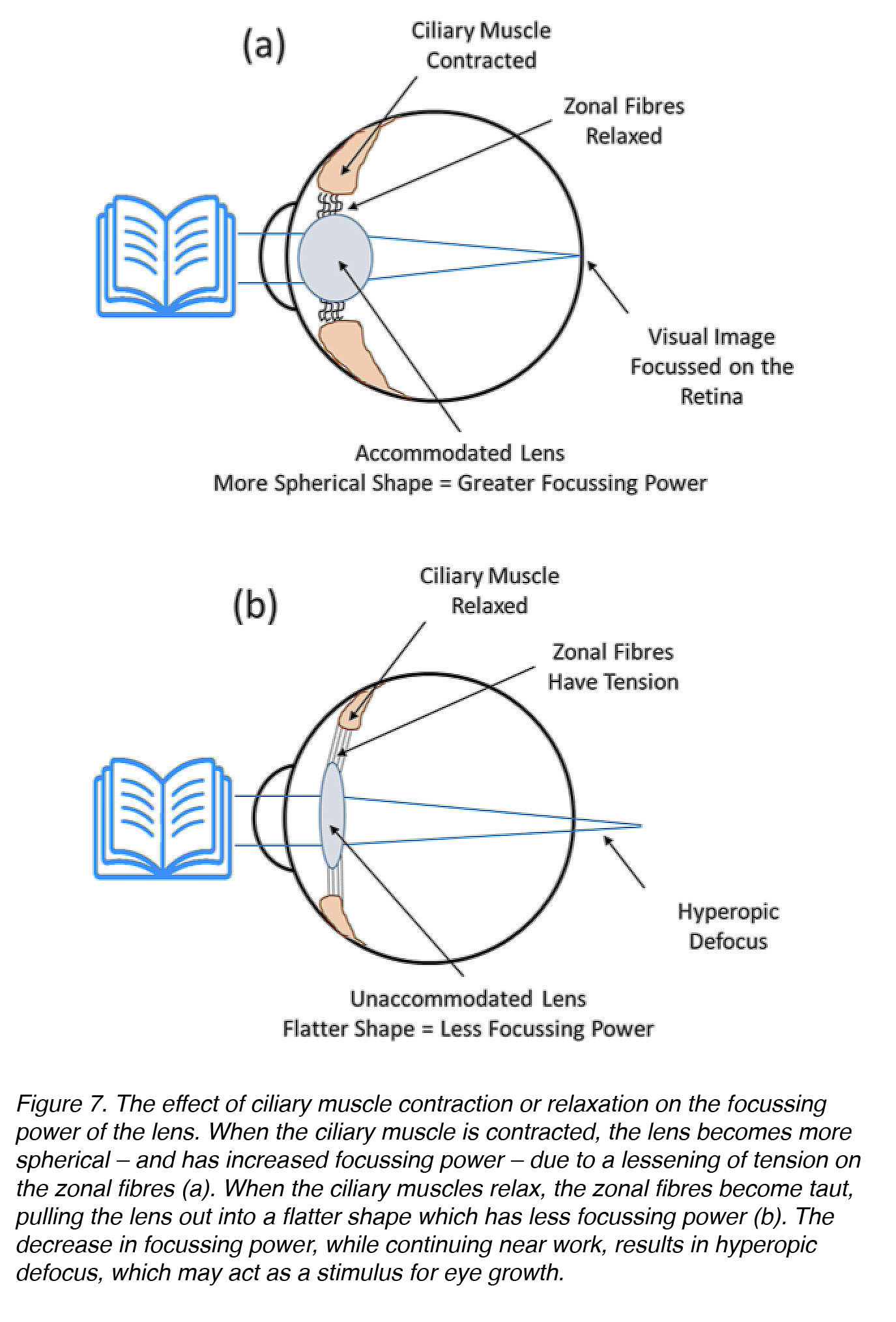
Perhaps you are familiar with the old wives’ tale, “Don’t sit too close to the TV, or you’ll ruin your eyes!”. This is likely because the most common and longstanding hypothesis about the cause of myopia development was that excessive near work results in accommodative fatigue, which then causes myopia. Accommodative fatigue occurs when the ciliary muscle weakens due to overexertion – as arm or leg muscles would – in response to an entire day of heavy lifting. Weakening of the ciliary muscle would result in loss of the focussing power of the lens, leading to hyperopic defocus when objects are viewed up close (Fig. 7). As discussed previously in the section about animal models of myopia, hyperopic defocus (pushing the plane of focus behind the photoreceptors) can be a stimulus for eye growth.
Figure 7. The effect of ciliary muscle contraction or relaxation on the focussing power of the lens. When the ciliary muscle is contracted, the lens becomes more spherical – and has increased focussing power – due to a lessening of tension on the zonular fibres (a). When the ciliary muscles relax, these fibres become taut – pulling the lens out into a flatter shape, which has less focussing power (b). The decrease in focussing power while continuing near-work results in hyperopic defocus, which, in principle, might act as a stimulus for eye growth.
To investigate whether reducing the need for accommodation in children would be preventative against myopia, scientists have utilized progressive-addition lenses (PALs). In principle, the use of PALs should allow accommodation (contraction of the ciliary muscle) to relax, while keeping a blur-free image on the retina [30] (Fig. 8).
Visual image focussed on the retina
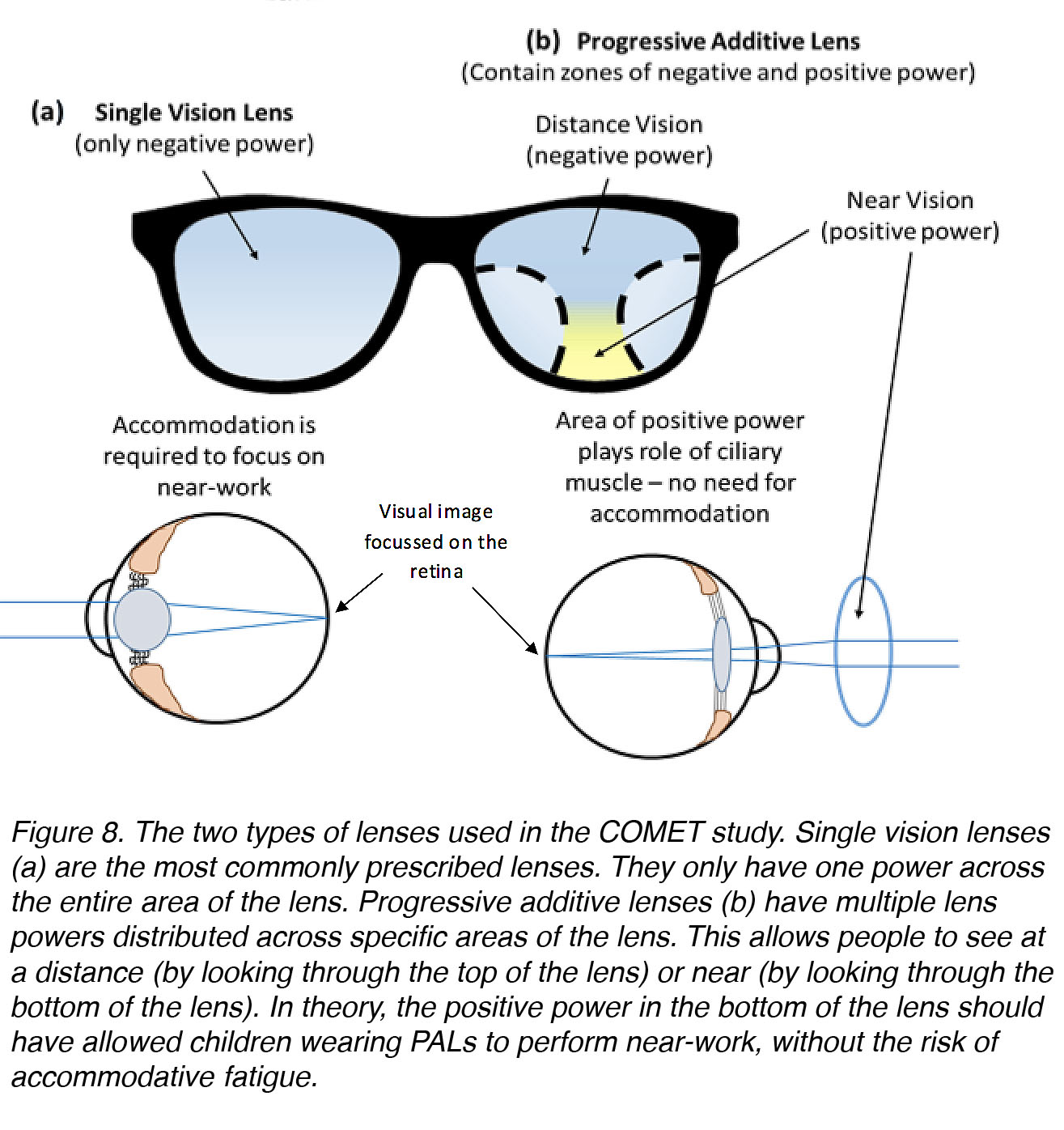
Figure 8. The two types of lenses used in the COMET study. Single vision lenses (a), which have only one power across their entire area, are the most common for correction of myopia. Progressive addition lenses (b) have multiple lens powers distributed across specific areas of the lens. This allows the wearer to focus at a distance (by looking through the top of the lens) or near (by looking through the bottom of the lens). In theory, the positive power in the bottom of the lens should have allowed children wearing PALs to perform near-work without the risk of accommodative fatigue.
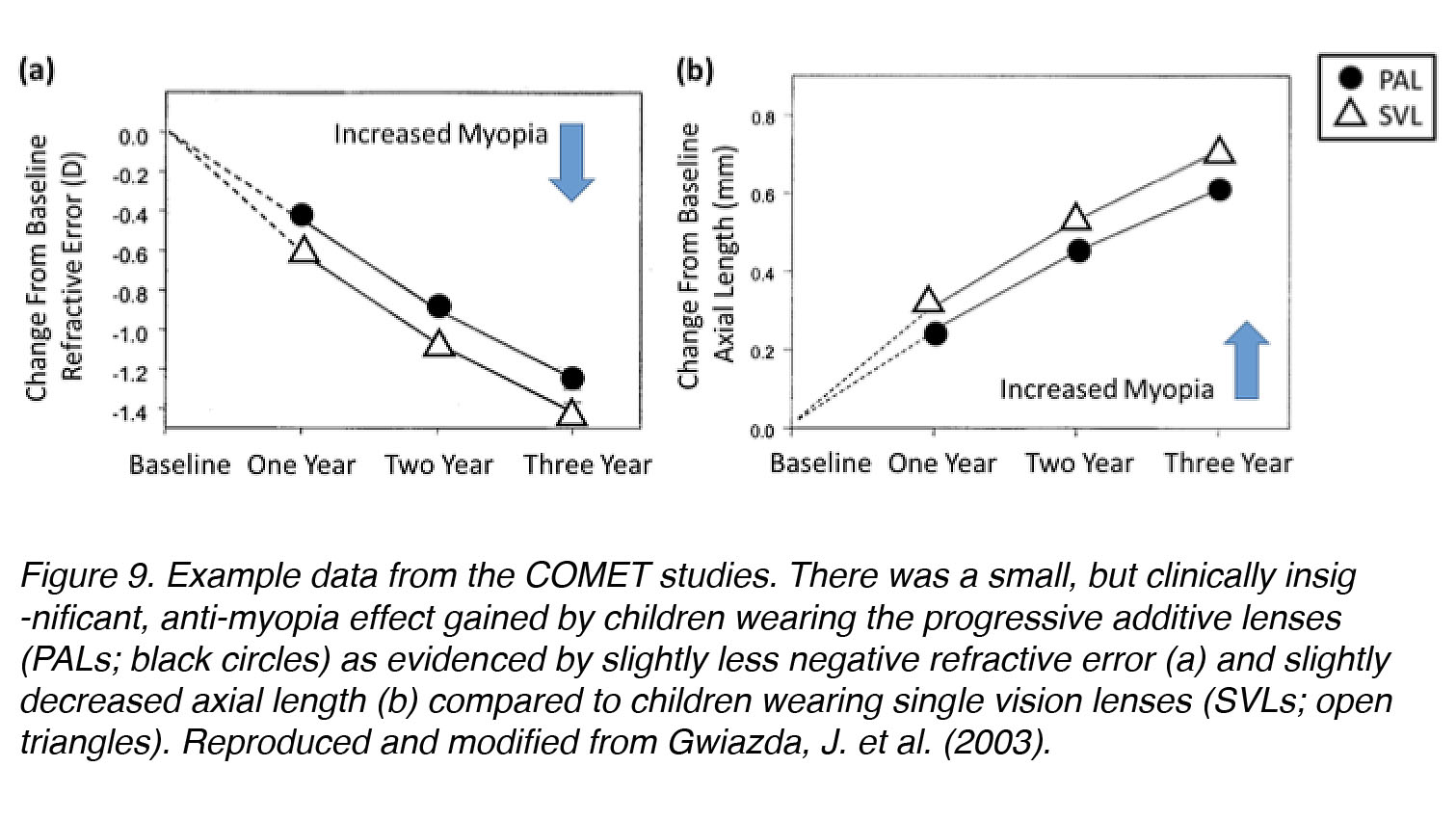
“The Correction Of Myopia Evaluation Trial” (COMET) was the largest and most significant human clinical trial ever conducted to test whether relaxation of accommodation through the use of PALs could help to inhibit the progression of myopia in children. It was a multicenter, randomized, double-masked, controlled clinical trial that took place over a period of three years (n = 469 children). At the end of the study, a statistically significant 3-year mean difference was found in children who wore PALs versus single vision lenses (SVL) (refractive error progression: PAL: −1.28 ± 0.06 D vs. SVL: −1.48 ± 0.06 D, p = 0.004; axial length elongation: PAL: 0.64 ± 0.02 mm vs. SVL: 0.75 ± 0.02 mm, p = 0.0002). These data supported the hypothesis that changes in accommodation can affect myopia progression. Unfortunately, this effect was too small to be considered clinically significant, although one subset of children – those who had high accommodative lag – seemed to benefit significantly from PAL wear (Fig. 9). Subsequent studies have confirmed these findings [31], reporting a statistically significant, but small, reduction in myopia progression in children wearing PALs versus SVLs; these effects were not dependent on accommodative lag, as high-lag children had similar results as others. The accumulation of experimental evidence from animal models and further human trials, however, does not support a significant role of accommodation in myopia development [32]. It remains possible that most PAL-wearers in the COMET study did not use the lenses properly, but tolerated the blur that resulted from accommodative fatigue and continued near-work without adjusting viewing angle for better focus.
Figure 9. Example data from the COMET studies. A small, but clinically insignificant anti-myopia effect was gained by children wearing the progressive additive lenses (PAL; black circles) as evidenced by slightly less negative refractive error (a) and slightly decreased axial length (b) compared to children wearing single vision lenses (SVL; open triangles). Reproduced and modified from Gwiazda, J. et al. (2003) [30].
A second hypothesis is that accelerated eye growth can occur because of over-correction of myopic refractive error (similar to lens-induced myopia). To test this hypothesis, children were under-corrected by +0.5 to +0.75 D. This trial therapy was largely unsuccessful; myopia progressed more rapidly in under-corrected children than in fully-corrected controls [33-34]. Interestingly, a recently published study [35] reported that myopia and axial elongation progressed more rapidly in fully under-corrected than in fully corrected myopic children. This held true even when controls were applied for other risk variables, such as baseline refractive error and axial length, number of myopic parents, age at myopia onset, and time spent doing near work and outdoors [35]. Thus, there is still no consensus on whether under-correction may be an effective anti-myopia therapy.
INTENSE OUTDOOR LIGHT PROTECTS AGAINST MYOPIA
In recent years, spending time outdoors has been reported to reduce the risk of developing myopia, and this effect does not seem to be dependent on the amount of physical activity [36-37]. We do not yet know the mechanisms through which outdoor light might protect against myopia, but some ideas are that it might (a) stimulate intensity- or wavelength-dependent anti-myopia systems in the retina, (b) cause sustained pupillary constriction via the melanopsin system – thus improving retinal image quality by reducing longitudinal aberrations, (c) increase the production of vitamin D in the skin, (d) increase the average viewing distance of objects outdoors compared to indoors, thereby reducing accommodative fatigue, or (e) increase the activation of spatiotemporal image-response mechanisms in the retina, which inhibit myopia development. For a recent review on the roles each of these factors may play in myopia, please refer to “The role of luminance and chromatic cues in emmetropization” by Frances Rucker [38]. For more information about the melanopsin system, refer to the Webvision chapter “Melanopsin-expressing, Intrinsically Photosensitive Retinal Ganglion Cells (ipRGCs)” by Dustin M. Graham and Kwoon Y. Wong.
High-Intensity Illumination
When Rose et al. studied the effects of time spent outdoors on the development of myopia in Australian children, they found that children who spent more time outdoors had lower associated myopic refractive errors than children who spent most of their time indoors, even after adjusting for near-work, parental myopia, and ethnicity [39]. Exercise versus leisure did not affect associated levels of myopia (Fig. 10).
Figure 10. Multivariable-adjusted odds ratios (adjusted for gender, ethnicity, parental myopia, parental employment, and education) for myopia development by reported average daily hours spent on near-work versus outdoor activities in 12-year-olds. Activities were divided into columns of high, moderate, and low levels. The group with high levels of outdoor activity and low levels of near work (striped blue arrow) is the reference group. The blue arrow denotes the group with high levels of near work and high levels of outdoors activity. Yellow and yellow-striped arrows denote high near work and low outdoors activity, and low near work and low outdoors activity, respectively. Reproduced and modified from Rose, K. A. et al. (2008) [39] .
Therefore, it was proposed that the higher mean illuminance (light intensity) outdoors – which can be as much as 100,000 lux in Sydney AU, compared to typical indoor values of 350-500 lux – was responsible for the protective effect of outdoor activity against myopia progression. Human pilot studies to test this hypothesis have been successful in inhibiting myopia, in a small cohort of Chinese schoolchildren [40] and in a larger cohort of Australian schoolchildren [37]. High-intensity light is also protective against experimentally-induced myopia in monkeys [41-42] and chicks [43-44]. The mechanism through which intense light may prevent myopia is unknown, but we can use animal models to investigate the signalling molecules that may be involved. There are substantial links to the up-regulation of neuromodulators such as dopamine [45] and nitric oxide [46]. These neurotransmitters are strongly correlated with myopia-inhibition on their own [47-48], and they may also work together in the same myopia-inhibiting pathway [49].
Wavelength
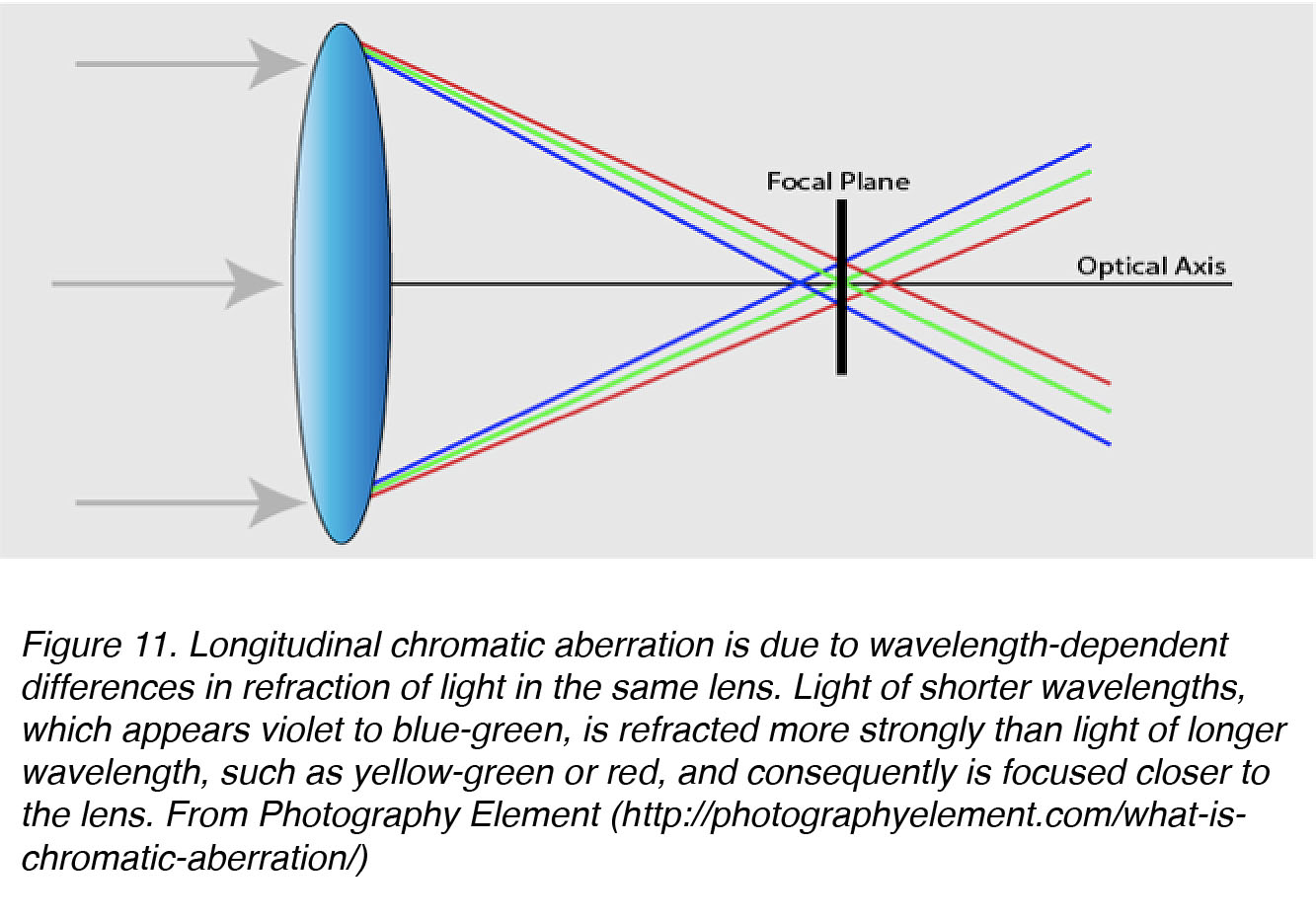
During emmetropization, the eye needs some sort of visual cue to determine the direction of blur [50], and chromatic aberrations have been suggested as a possible cue [38]. The most common chromatic aberration is longitudinal chromatic aberration (LCA), in which different wavelengths of light are focussed at different distances along the same axis (Fig. 11).
Figure 11. Longitudinal chromatic aberration is due to wavelength-dependent differences in refraction of light by the same lens. Light of shorter wavelengths, which appears violet to blue-green, is refracted more strongly than light of longer wavelength, such as that which appears yellow-green or red, and consequently is focused closer to the lens. From Photography Element (http://photographyelement.com/what-is-chromatic-aberration/)
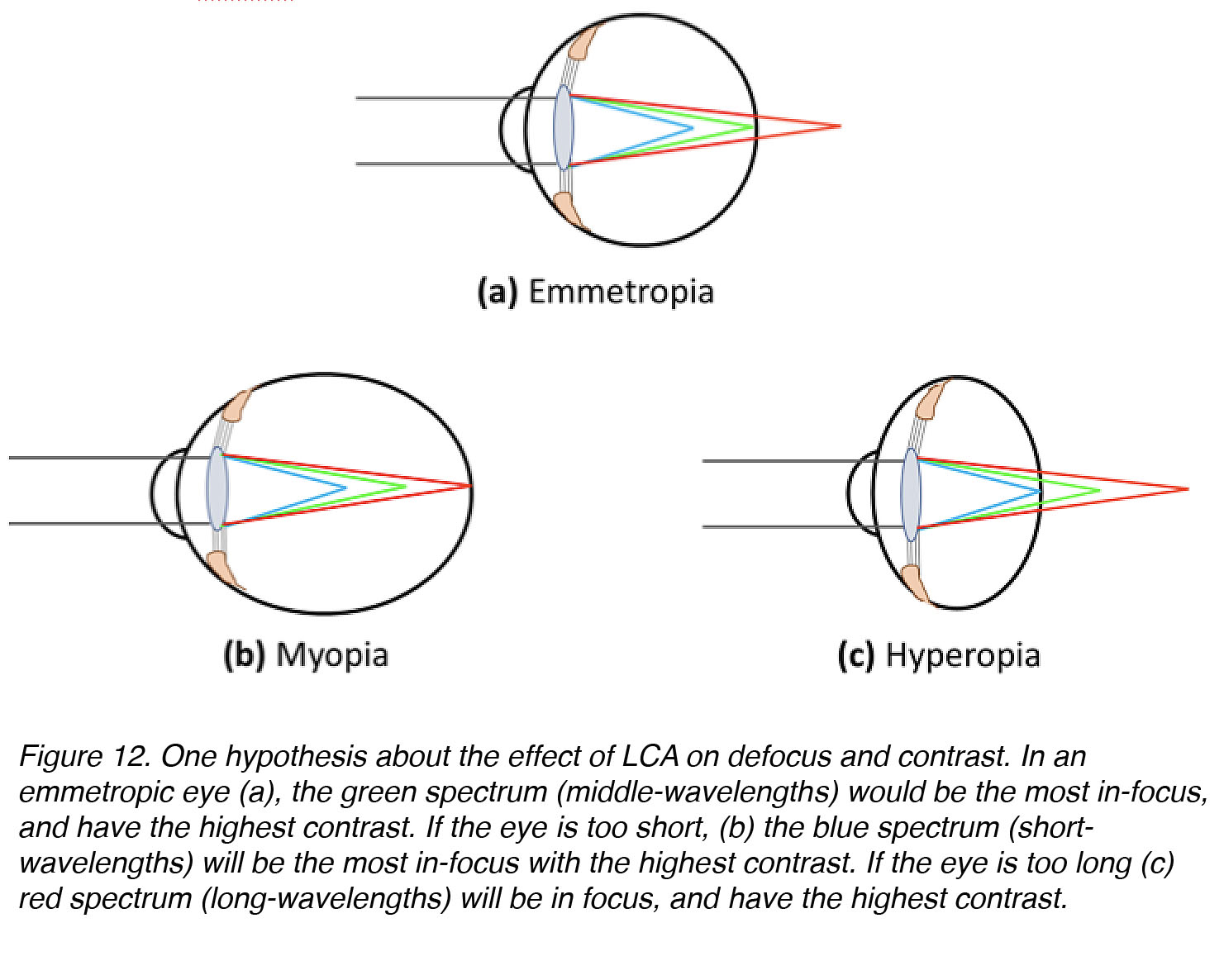
In humans, the fovea is most densely populated with medium- and long-wavelength-sensitive cones. One idea is that the eye should be guided to grow to a size at which middle wavelengths in the visible spectrum (yellow-green, 550-570nm) will be the most sharply focussed, with the highest contrast, to attain the highest possible spatial acuity. If this were the case, the in-focus image of an eye that is too small will be blue-shifted, with high contrast at short-wavelengths (450-470 nm), whereas the in-focus image of an eye that is too large will be red-shifted with high contrast at long-wavelengths (610-630 nm) (Fig. 12).
Figure 12. One hypothesis about the effect of LCA on defocus and contrast. In an emmetropic eye (a), the green region of the spectrum (middle-wavelengths) would be the most in-focus and have the highest contrast. If the eye is too short (b), the blue region of the spectrum (short-wavelengths) will be the most in-focus and have the highest contrast. If the eye is too long (c), the red region spectrum (long-wavelengths) will be most in-focus and have the highest contrast.
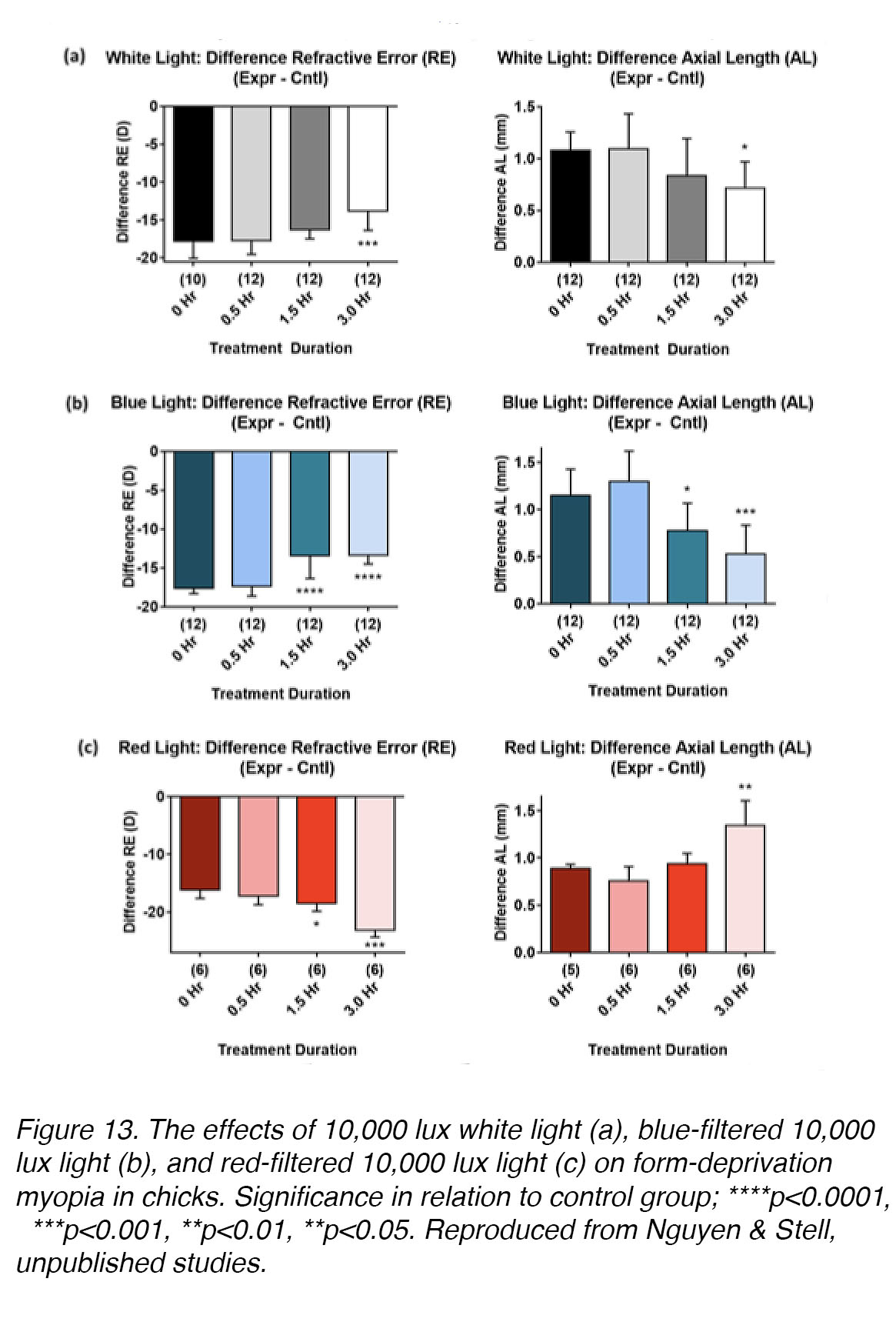
The results of research on the effects of LCA on emmetropization in the eye show that it is sufficient to detect the direction of retinal blur [51], but it does not seem to be necessary for emmetropization [52-54]. Despite this, many studies have demonstrated clearly that the chromaticity (spectral composition) of light can be an important modulator of eye growth, although its effects do not seem to be the same in all animal models. In chickens raised without goggles or lenses, short-wavelength (blue) light retards eye growth, whereas long-wavelength (red) light promotes it; white light (which contains all spectral wavelengths) has no effect [55]. In chicks wearing monocular diffusers, long-wavelength illumination (achieved using filters) is myopia-promoting, while short-wavelength illumination is myopia-inhibiting (Ghodsi & Stell [56], and Nguyen & Stell, unpublished studies; Fig. 13). Form-deprivation myopia in guinea pigs also responds to narrow-band long- or short-wavelength exposure in the same way as chicks [57-59]. In contrast, long-wavelength light is growth-inhibiting and short-wavelength light is growth-promoting in rhesus monkey [60] and tree shrew (Gawne et. al. [56]). Thus, further investigations are needed to explore the effects of light spectrum on myopia development and to identify the underlying retinal mechanisms, in various animal models.
Figure 13. The effects of 10,000 lux white light (a), blue-filtered 10,000 lux light (b), and red-filtered 10,000 lux light (c) on form-deprivation myopia in chicks. Significance in relation to control group; ****p<0.0001, ***p<0.001, **p<0.01, **p<0.05. Reproduced from Nguyen & Stell, unpublished studies.
Retinal circuitry and ocular pathways of myopia
Emmetropization is an active-feedback process, guided by visual cues. It aims to match the axial length and resting focal power of the eye in such a way that images of objects viewed at any distance, can be focussed onto the retinal photoreceptors by accommodation [61].
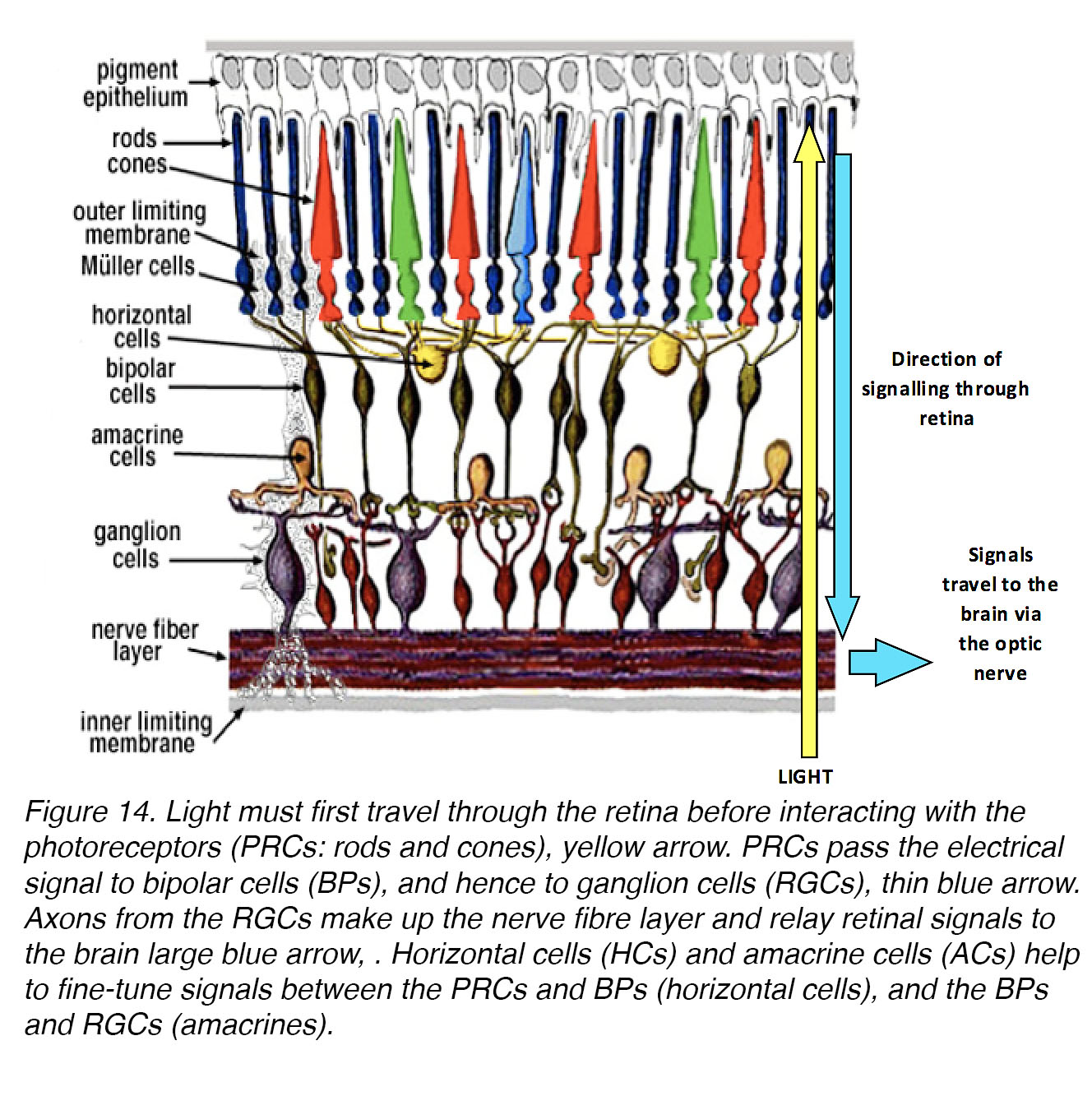
Past studies indicate that the retina is the primary source of signals that regulate eye growth19, while pathways involving transmission between retina and brain, via the optic nerve, may play roles in focal refinement of the visual image [62]. The retina is a highly specialized neural tissue that lies at the back of the eye (Fig. 14).
Figure 14. Light must first travel through the retina before interacting with the rod and cone photoreceptors (PRs) – yellow arrow. PRs signal their responses via synapses to bipolar cells (BCs) and hence to ganglion cells (RGCs) – thin blue arrow. Axons from the RGCs make up the nerve fibre layer, and relay retinal signals to the brain, large blue arrow. Horizontal cells (HCs) and amacrine cells (ACs) help to fine-tune signals – between the PRs and BCs, and between the BCs and RGCs, respectively.
Although evidence suggests that the retina is the source of ocular growth-regulating signals19, we do not know the specific cell types involved in emmetropization. There is no evidence for the direct involvement of horizontal cells, but destruction of photoreceptors (PRs) and/or amacrine cells (ACs) in the chick retina has been associated with increased axial growth of the eye [63-64]. Given the broad-spectrum action of toxins on retinal neurons, the relative roles of PRs and ACs in myopia-control in these types of studies are not well-defined, and molecular tools which would disrupt function of retinal cell types more specifically than toxins, are not widely-researched or available in chicks, the most common myopia model organism. Animals in which molecular tools are currently available – such as mice – are less suitable for studies of eye growth because their eyes are extremely small. This makes it very difficult to measure accurately any changes in axial growth or refractive error. This drawback of the chick model may change with a new genome-editing technology called CRISPR [65-66], which is an exciting development in the myopia-research field. Delivery of gene-silencing agents to cells in the retina of hatched chicks presents challenges that we have still not overcome (Waldner D, et al. IOVS 2017; [58]: ARVO E-Abstract 5905).
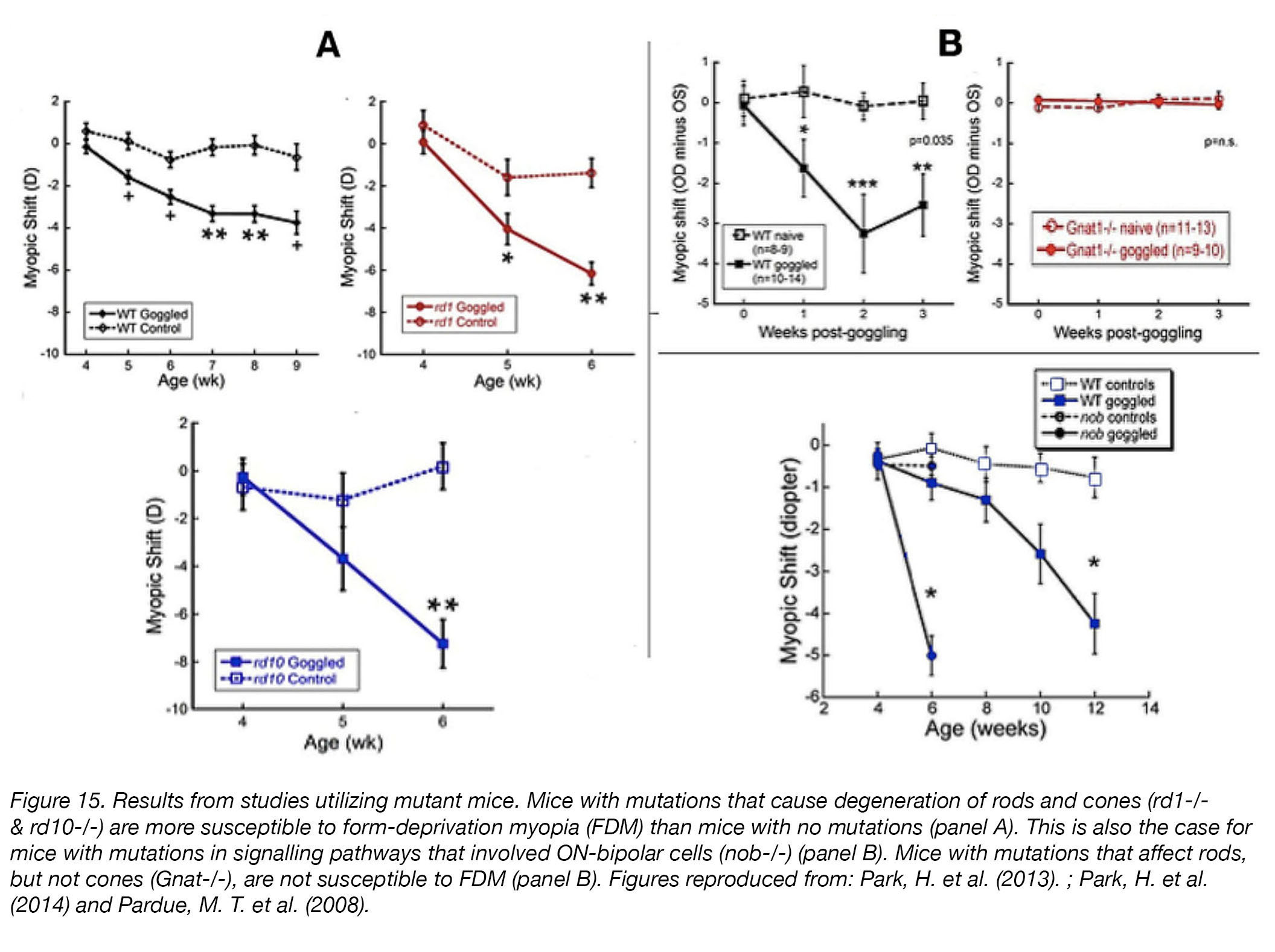
That is not to say that myopia experiments in mice have not been performed. Genetic “knockout” mice models – in which their genome has been edited to “remove” certain retinal cell types or visual processing mechanisms by causing mutations – were used to determine the contributions of rods, cones, and retinal ON-pathways (some bipolar cells) to emmetropization [67]. Eyes of mice with mutations causing severe degeneration of rods and cones (rd1-/- & rd10-/-) were more susceptible to form-deprivation myopia than those of their wild-type (unmanipulated) counterparts [68] (Fig. 15), while the eyes of rod-specific knockout Gnat1-/- mice, which exhibit little retinal damage, were unresponsive to form-deprivation [69]. Eyes of mice with a mutation in nyctalopin (Nyxnob/nob) – a protein important in ON-bipolar cell signalling – exhibited an increased susceptibility to form-deprivation myopia [70].
Figure 15. Results from studies utilizing mutant mice. Mice with mutations that cause degeneration of rods and cones (rd1-/- & rd10-/-) are more susceptible to form-deprivation myopia (FDM) than mice with no mutations (panel A). This is also the case for mice with mutations in signalling pathways that involved ON-bipolar cells (nob-/-) (panel B). Mice with mutations that affect rods, but not cones (Gnat-/-), are not susceptible to FDM (panel B). Figures reproduced from: Park et al. (2013) [68]; Park et al. (2014) [69]; Pardue et al. (2008) [70].
This led the authors to conclude that perhaps rod-mediated signalling in the retina is more important for emmetropization than cone-mediated signalling. This revolutionary concept is supported further by the fact that rod photoreceptors signal primarily through ON-bipolar cells, which likely mediate the effects of the injected glutamate analog, L-APB, on eye growth in chicks [71]. Furthermore, rods vastly outnumber cones in most mammalian retinas, including those of mice and humans; and vision with high spatial resolution, which is not mediated by rods, is not necessarily required for the retina to successfully detect blur [19-72]. It is possible that the transgenic ablation of rod-ON-BCs could have damaged the inner-retinal circuitry that produces myopia-STOP signals, and the ability of mice with intact rods, but no cones, to develop FDM has not yet been investigated. Thus, we would hope to see further evidence in support of this counter-intuitive hypothesis.
Ganglion cells, and therefore cortical visual pathways, do not seem to contribute significantly to homeostatic eye growth, even though ganglion cell axons make up the optic nerve. Disconnection of the optic nerve – either by surgery or toxins – has little effect on emmetropization and/or compensation to induced defocus26, [73-75], and eyes will still respond to deprivation myopia [26]. Retinal efferent (centrifugal) fibres also run through the optic nerve, in birds and other animals. Unilateral lesioning of this system, while leaving retinal ganglion cell axons intact, results in hyperopia in the eye contralateral to the disrupted fibre tract. By 21 days after surgery, however, the eyes return to an emmetropic state [76] – suggesting, perhaps, that the initial hyperopic shift was due to short-term release of neuroactive substance(s) such as NO from the retinal axon terminals of the damaged cells, or retrograde signalling within the retina by axotomized RGCs. There is a precedent for a process such as this in the induction of rod-precursor proliferation in goldfish, which can be elicited by sectioning of the optic nerve and ablation of olfactory bulb/tract – the source of efferent fibres in fish [77]. Many studies do not report intra-ocular effects of treatment, even though direct retino-retinal connections have been shown to exist in rats and mice [78-79].
Amacrine cells are the visual processing powerhouses of the retina, and the most likely source of retina-derived eye growth-regulating molecules [19]. There are as many as 30 different confirmed subtypes of amacrine cells, and it is likely that more remain to be discovered [80-81]. The majority of amacrine cells provide inhibitory feedback to bipolar cells via GABA- and glycine-mediated signalling; but they can also contact and provide feedback to other amacrine cells and ganglion cells [81-82], as well as release neuromodulatory substances that modify properties such as excitability, gain, synaptic transmission, and cell-cell coupling of many retinal neurons and circuits, through non-synaptic mechanisms. They are responsible for higher-level processes in the retina such as detecting environmental changes in movement [83], contrast, and blur [84]. For more information on amacrine cell function, see the Webvision chapter “Roles of Amacrine Cells” by Helga Kolb.
Many studies have shown that various signalling molecules found in amacrine cells can affect myopia onset and inhibition [47,85-89], such as dopamine [47], nitric oxide [48], and neuropeptides (VIP, glucagon, somatostatin, and neurotensin) [80]. Destruction of subpopulations of amacrine cells by toxins has significant effects on normal eye growth and induction of form-deprivation myopia [63,64,90-92]. The functions of most amacrine cells identified remain to be discovered, but the contribution of specific amacrine cell-derived signalling molecules to emmetropization and myopia inhibition will be discussed in more detail below. Most likely, signalling molecules originating from the retina do not control eye growth directly; instead, they are assumed to act via a signalling cascade that relays eye growth-regulating signals through the RPE and choroid, which may then release different signalling molecules that directly affect scleral growth [19] (Fig. 16).
Figure 16. It is not likely that signals from the retina will control scleral growth directly. Instead, retina-derived chemical messengers probably activate signalling cascades that create a chain reaction of signalling, which moves through the RPE, choroid and sclera (arrows). For example, dopamine and/or nitric oxide signalling in the retina can cause changes in light adaptation, which might then result in the release of retinoic acid from the choroid. Choroidal retinoic acid release could then result in inhibition of scleral proliferation, and inhibition of myopia. Although all the molecules listed in the diagram have been shown to affect eye growth, the places where they may act are still theoretical, and have not yet been proven conclusively. Abbreviations: TGF-β, transforming growth factor beta; bFGF, basic fibroblast growth factor; mAChRs, muscarinic acetylcholine receptors; NO, nitric oxide; DA, dopamine; EGR-1, early growth response protein 1 (also known as ZENK); ACh, acetylcholine; GABA, gamma-aminobutyric acid.
MUSCARINIC ACETYLCHOLINE RECEPTORS, DOPAMINE, AND NITRIC OXIDE ARE STRONGLY IMPLICATED IN REGULATION OF EYE GROWTH
Muscarinic Acetylcholine Receptors (mAChRs)
Muscarinic receptors in the eye mediate contraction of the pupillary constrictor muscle, as well as the ciliary muscle, which controls accommodation. As discussed previously, it was long thought that myopia inhibition could be achieved by blockade of accommodative fatigue. However, the failure of clinical trials such as COMET (discussed above), plus experimental evidence from animal models, does not supported the hypothesis that mAChR antagonists (blockers) inhibit myopia via paralysis of the ciliary muscle alone32. Chicks can develop experimentally-induced myopia and/or respond to treatment with drugs such as atropine or pirenzepine – which block muscarinic receptors [93-94] – even though their ciliary muscles are controlled by nicotinic receptors, which atropine and pirenzepine do not target. Pirenzepine inhibits the progression of myopia in humans [95-97] and a wide range of animal species [25] including monkey [98-99], tree shrew [100-102], and chick [94,103-105], even though it has little effect on iris and ciliary musculature [99,102,103,106,107]. Finally, lesioning of the ciliary nerve does not impair development of either form-deprivation or lens-induced myopia; chicks with damaged ciliary nerves develop similar amounts of myopia as intact controls [108].
These data have largely ruled out accommodative activity or the ciliary muscle as an important eye-growth-regulator; thus, we have turned to the retina, choroid, or sclera as possible target sites for myopia-inhibition by mAChR antagonists. Muscarinic receptors are present in the retina, RPE, choroid, and sclera [109]. Fischer et al. localized chick mAChR orthologues (cM2, cM3, cM4) in chick eye tissues and showed that immunoreactive sites are present in the inner retina (cholinergic amacrine cells and ganglion cells), retinal pigment epithelium (RPE), and choroid, as well as the ciliary body [110]. Expression patterns in the tree shrew [111], and rabbit [112] are similar to those reported for the chick (Fig. 17). However, even though there is evidence for mAChRs in the ocular tissues, it remains uncertain exactly which ocular tissues, cells, or receptors mediate the anti-myopia effects of mAChR antagonists. The theorized mechanism of action of myopia-inhibition by these drugs is heavily dependent on the assumption that they block signalling through mAChRs, but this has never been proven conclusively.
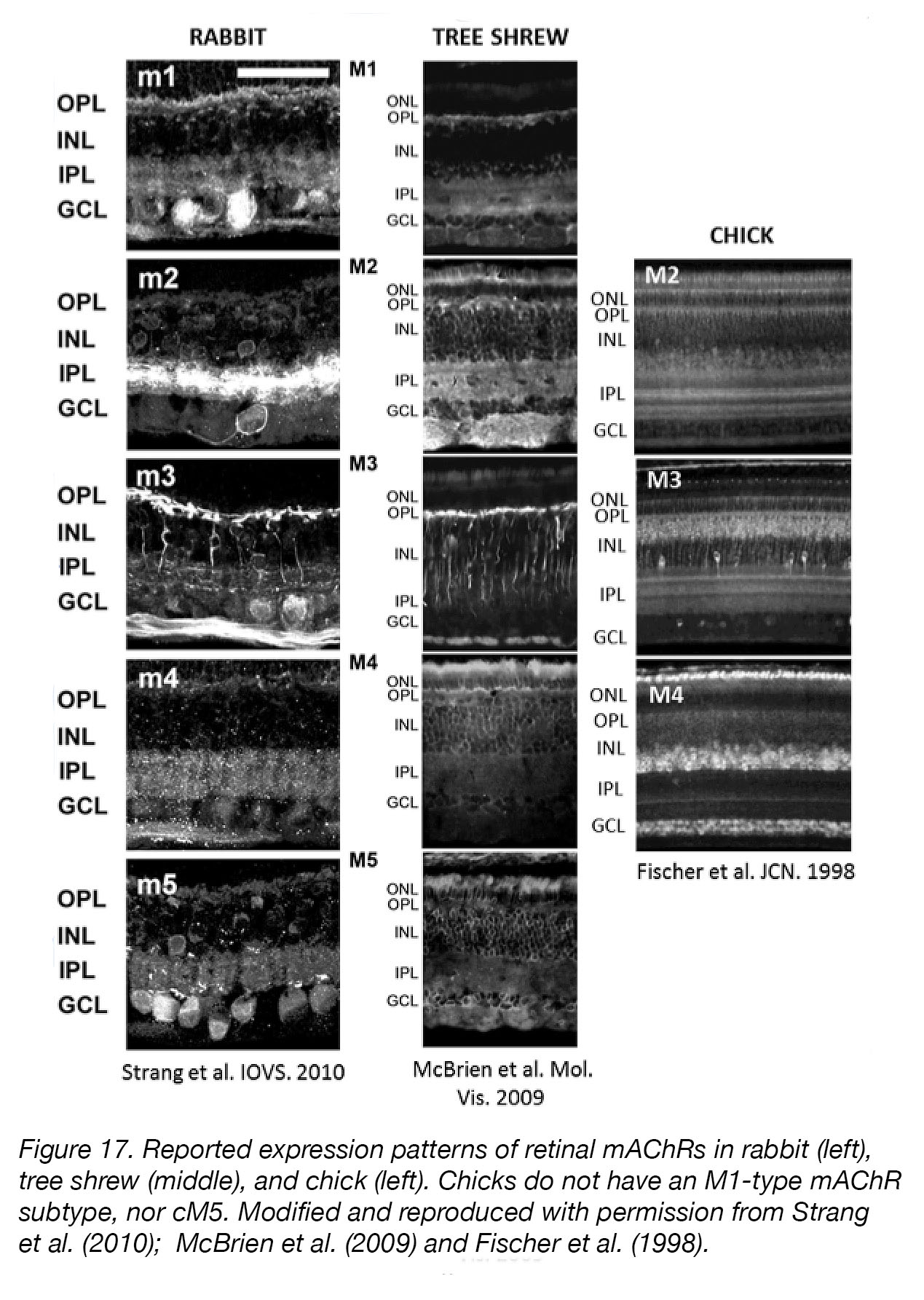
Figure 17. Reported expression patterns of retinal mAChRs in rabbit (left), tree shrew (middle), and chick (left). Chicks do not have an M1-type mAChR subtype [113]. Modified and reproduced with permission from Strang et al. [112]; McBrien et al. [111] and Fischer et al. [110]
In fact, substantial evidence argues for a non-retinal or non-muscarinic mode of action [114]. For example, only some muscarinic antagonists inhibit myopia in humans [95,96,115-117] and animals [101,104,118], while the vast majority have no effect [104]; high concentrations of these drugs are required for a significant effect. Protein and mRNA expression studies utilizing radioligand binding and real-time PCR techniques have found no change in receptor density or receptor gene expression for any mAChR subtypes in the retina or choroid of chick [119] or tree shrew [111], even when there is significant myopia development. This is strange, as it would be expected that significant changes in eye growth should cause changes in the expression of target receptors. Interestingly, amacrine cells that make the neurotransmitter required for action by mAChRs (acetylcholine) do not seem to be required for emmetropization, development of experimentally-induced myopia, or atropine-mediated suppression of form-deprivation myopia [120]. This is again very strange, because ‘orthosteric’ mAChR antagonists such as atropine should have no action by themselves; they simply block access of the signalling neurotransmitter to the target receptor. Thus, if myopia inhibition by atropine is mediated by mAChRs, atropine should not inhibit myopia if the source of acetylcholine has been destroyed.
These data support a few possible theories: The first is that the cells responsible for myopia inhibition by mAChR antagonists are not in the retina, but instead in the RPE, choroid, or sclera. When muscarinic antagonists are administered by injection into whole, intact eyes, this is a possibility. For a drug to have a significant effect on a system, it must be present in a certain concentration at the receptor for binding and activation to occur. The inverse of this effective concentration is called ‘potency’. Atropine is a very potent drug; thus, it requires an extremely low concentration to block mAChR signalling (1-10 nM). If it were working at a target far away from the intravitreal injection site (i.e., the choroid or sclera), the requirement for a large concentration of drug could be justified; it is possible that only a small amount of atropine might reach its target, through loss by diffusion through the vitreous, and/or blockade by the RPE and blood-retinal barrier. If the target tissues (retina, choroid, sclera) are isolated, however, the requirement for large concentrations of drug should be eliminated, because these barriers have been removed. One study examining the effects of mAChR agonists (stimulators) and antagonists on eye cups consisting of the RPE/choroid/sclera (in vitro; retina removed) demonstrated that stimulation of mAChRs by carbachol, oxotremorine, arecadinine, and pilocarpine resulted in choroidal thinning, and that pirenzepine caused choroidal thickening; atropine was not tested, and oxyphenonium had no effect [121]. Another study on the effects of atropine on isolated scleral cells found that it can slow proliferation, which would be expected if atropine inhibited myopia by directly affecting scleral growth. Unfortunately, however, because the concentrations of drugs used in these isolated tissues were very high (0.6-5 mM), neither study conclusively indicates a non-retinal origin for mAChR-control of eye growth control.
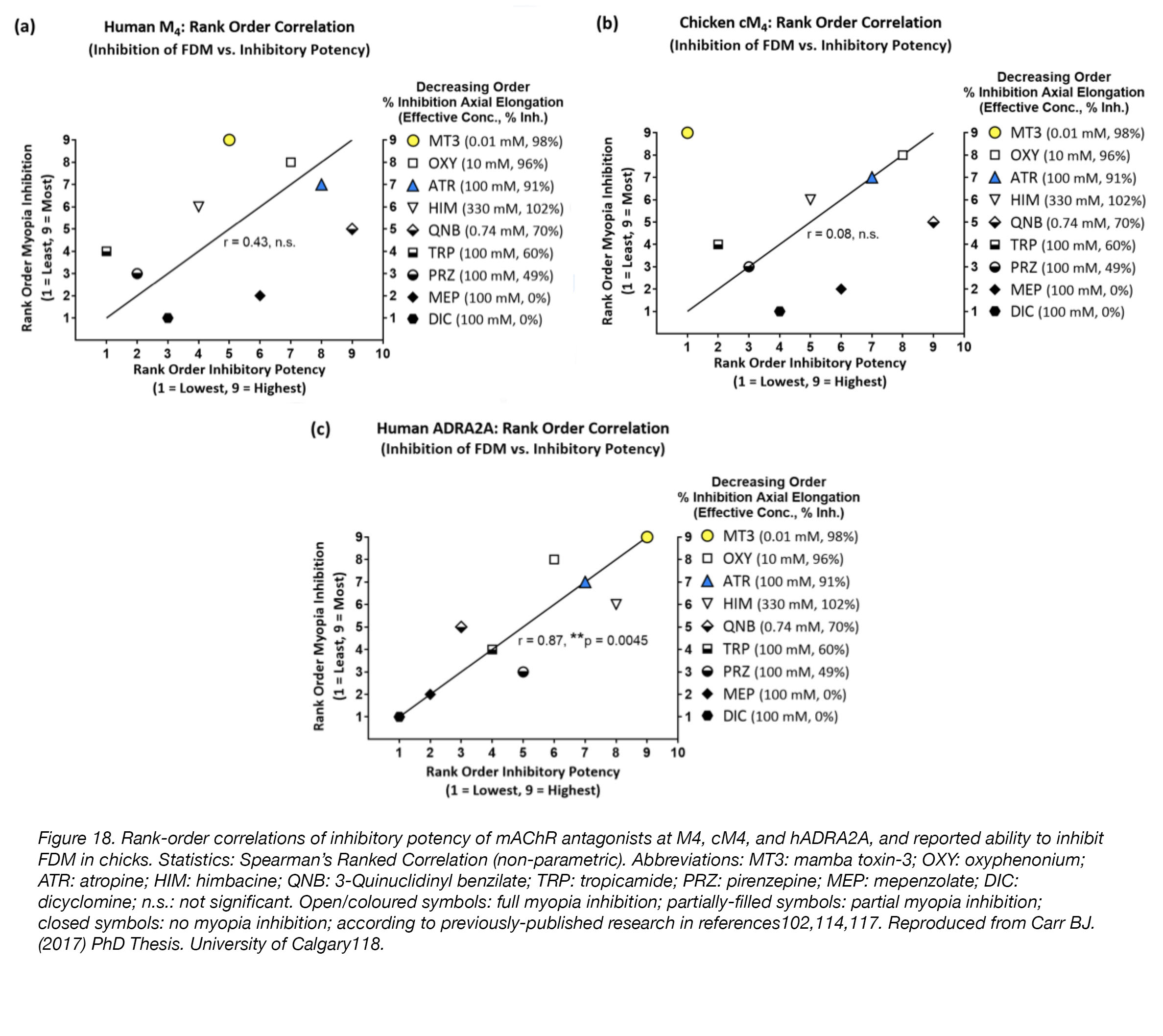
A second possibility is that high concentrations of mAChR antagonists are required for myopia inhibition because these drugs are not acting at mAChRs, but instead at some other, unknown receptor. All drugs are “dirty”, or promiscuous, meaning they can, and will, bind to unintended receptors; the risk of this “off-target” or “non-specific” binding increases as the concentration of drug increases. In the case of myopia-inhibition, extremely high concentrations – up to one-million times greater than those required for binding (1 mM used vs. 1 nM required; see above) – are used routinely. Thus, it’s no great stretch of the imagination to speculate that when drugs are used at concentrations such as these, they will act in ways we cannot accurately predict. To determine whether “off-target” binding of mAChR antagonists is possible, we conducted in vitro experiments using isolated human mAChR M4 receptor (M4), chicken mAChR M4 receptor (cM4), and human alpha2A-adrenoceptor (hADRA2A). Our hypothesis was that myopia-inhibiting mAChR antagonists would bind to hADRA2A – an “off-target” receptor – when applied at high concentrations. We found that myopia-inhibiting mAChR antagonists bind to hADRA2A when applied at concentrations similar to those expected to be present when these drugs are used to treat myopia in the chick model (0.001-1 mM)104, while mAChR antagonists that do not inhibit myopia did not bind to this receptor with any significant activity, or at all. Interestingly, the correlations between the ability of mAChR antagonists to bind to M4/cM4 receptors and their reported ability to inhibit myopia in chick [104] were very poor, whereas correlations between mAChR antagonist binding at hADRA2A and myopia inhibition in chick were significantly better (Fig. 18).
Figure 18. Rank-order correlations of inhibitory potency of mAChR antagonists at M4, cM4, and hADRA2A, and reported ability to inhibit FDM in chicks. Statistics: Spearman’s Ranked Correlation (non-parametric). Abbreviations: MT3: mamba toxin-3; OXY: oxyphenonium; ATR: atropine; HIM: himbacine; QNB: 3-Quinuclidinyl benzilate; TRP: tropicamide; PRZ: pirenzepine; MEP: mepenzolate; DIC: dicyclomine; n.s.: not significant. Open/coloured symbols: full myopia inhibition; partially-filled symbols: partial myopia inhibition; closed symbols: no myopia inhibition; according to previously-published research [104,118,122]. Reproduced from Carr BJ. (2017). PhD Thesis. University of Calgary [123].
Of additional importance were our findings regarding the experimental anti-myopia ligand, MT3, a polypeptide isolated from venom of the green mamba snake. Recently, it has been reported that MT3 inhibits myopia in chicks at concentrations significantly lower than those required for atropine (2.5-10 μM vs. 1-100 mM, respectively) [122,124]. MT3 is known to be selective for mammalian mAChR M4 over all other mAChR subtypes [125]. Thus, the authors of these studies claimed that because MT3 inhibits myopia at a much lower concentration than atropine, mAChR M4 must be the “myopia-controlling” receptor. However, in our studies, we found that MT3 is the least potent antagonist of all tested at the chicken (cM4) receptor – 56x less potent than at human M4 (Fig. 19). This disconnect between MT3-potency at cM4 and its rank as the most potent anti-myopia ligand in the chick – and the fact that MT3 binds with high potency, not only to mAChR M4, but also to alpha1A-, alpha1D-, and alpha2A-adrenoceptors [126,127] – casts further doubt on the theory that M4/cM4 are the target receptors that mediate inhibition of myopia by muscarinic antagonists.
Figure 19. Inhibition curves for MT3 at the human (M4) and chicken (cM4) muscarinic receptors. MT3 is 56x less potent at cM4 than M4 at the 50% effective inhibitory dose (dotted lines, IC50), effectively becoming a low-potency ligand at the chicken receptor; ****p<0.0001, unpaired t-test, two-tailed. Reproduced from Carr BJ. (2017). PhD Thesis. University of Calgary [123].
These data are preliminary, however, and should be interpreted with caution. They do not prove a role for alpha2A-adrenoceptors in control of eye growth. Rather, they disprove M4/cM4 as a likely target, and offer alpha2A-adrenoceptors as one possible alternative out of many – including other subtypes of mAChRs. There is some evidence for a role of alpha-adrenoceptors and their neurotransmitters, norepinephrine (NE) and epinephrine (E), in the eye. NE and E are the primary neurotransmitters in the sympathetic nervous system, from which the superior cervical ganglion originates. This ganglion projects to the eye and controls processes such as pupil dilation via the iris dilator papillae, reduction of ocular blood flow via vasoconstriction, and regulation of intraocular pressure via control of aqueous humor outflow [128]. A few studies have localized alpha-adrenoceptors in these ocular tissues, which is not surprising, but these receptors also seem to be present in the retina [129,130]. One hypothesized role of alpha2-adrenoceptors in the retina is that they might be involved in regulation of dopamine synthesis [131-133]. There is strong evidence that dopamine is a very important molecule in myopia-inhibition and regulation of eye growth (see below). Confirming these studies, and expanding upon them, may help to refine whether there is alpha2-adrenoceptor-mediated signalling in the retina, whether it can affect retinal dopamine regulation, and possibly identify cell types that might be involved. Confoundingly, although there is evidence for the presence of alpha2-adrenoceptors and the enzyme phenylethanolamine N-methyltransferase (PMNT) – which converts NE into E – in the retina, there is no significant evidence that either NE or E act as a retinal neurotransmitter [133]. For now, the function and activating transmitters of the alpha-adrenoceptors in the retina remains largely unknown. Significant further investigation will be required to determine whether these receptors are a worthy target for anti-myopia therapies.
Dopamine
Dopamine is perhaps one of the most-studied neurotransmitters in regards to regulation of eye growth. For an in-depth review of the studies investigating the effects of dopamine and dopaminergic systems on myopia, please see “An updated view on the role of dopamine in myopia” by Feldkaemper and Schaeffel [47] and “Dopamine signaling and myopia development: What are the key challenges” by Zhou et al. [134].
Dopamine is a catecholamine neurotransmitter/neuromodulator that acts as a light-adaptive signalling molecule in the retina [135]. It is synthesized in and released from tyrosine hydroxylase-containing amacrine cells. Intravitreal injection of dopamine agonists is strongly correlated with inhibition of eye growth [47,88,136-144] (Fig. 20), while injection of dopamine antagonists before unrestricted vision prevents myopia rescue [145].
Figure 20. The non-specific dopamine-receptor agonist (activator), apomorphine, inhibits form-deprivation myopia in the chick when injected via intravitreal and subconjunctival routes. Reproduced and modified with permission from Rohrer, B., Spira, A. W. & Stell, W. K. (1993) [136].
In addition to a direct role of dopamine agonism on inhibition of myopia in chicks, retinal dopamine levels and subsequent effects on myopia are sensitive to modulation by other drugs. Myopia-prevention by the cholinomimetic agent diisopropylfluorophosphate [146] and mamba toxin-3 in chick [147], and mamba toxin-7 (MT7) in tree shrew [148], can be blocked by dopamine antagonism. Interestingly, intravitreal injection of atropine or in vitro application of atropine to the chick retina causes increased levels of dopamine metabolite DOPAC and retinal dopamine release [141], and like atropine [48], myopia-inhibition by dopamine is blocked by simultaneous injection of nitric oxide synthase inhibitors [49,149]. Interestingly, the effects of dopamine agonists and muscarinic antagonists are not additive [142]. This led the authors to suggest that acetylcholine and dopamine may act at different points of a common pathway to prevent myopia; further investigation into this hypothesis has not been undertaken.
Nitric Oxide
Nitric oxide (NO) is a small-molecule gas that can act as a non-conventional neurotransmitter and neuromodulator in the retina. It is difficult to image NO directly, because it is volatile and chemically unstable. Consequently, past studies utilized histochemical activity of the enzyme NADPH-diaphorase [150] and antibody-labelling of NO-synthesizing enzymes (nitric oxide synthase, NOS) to determine the localization of NO action in tissues. NADPH-diaphorase activity and immunoreactive neuronal NOS (nNOS, NOS-1) are found in subpopulations of all major cell types in the chick retina and choroid, and at least 15 types of identified retinal neurons [151,152]. Direct imaging of NO has been accomplished by means of an indicator molecule that fluoresces after reacting with NO [153], which has been used to refine further the localization of various NOS isoforms, and where NO may act in the retina [154,155].
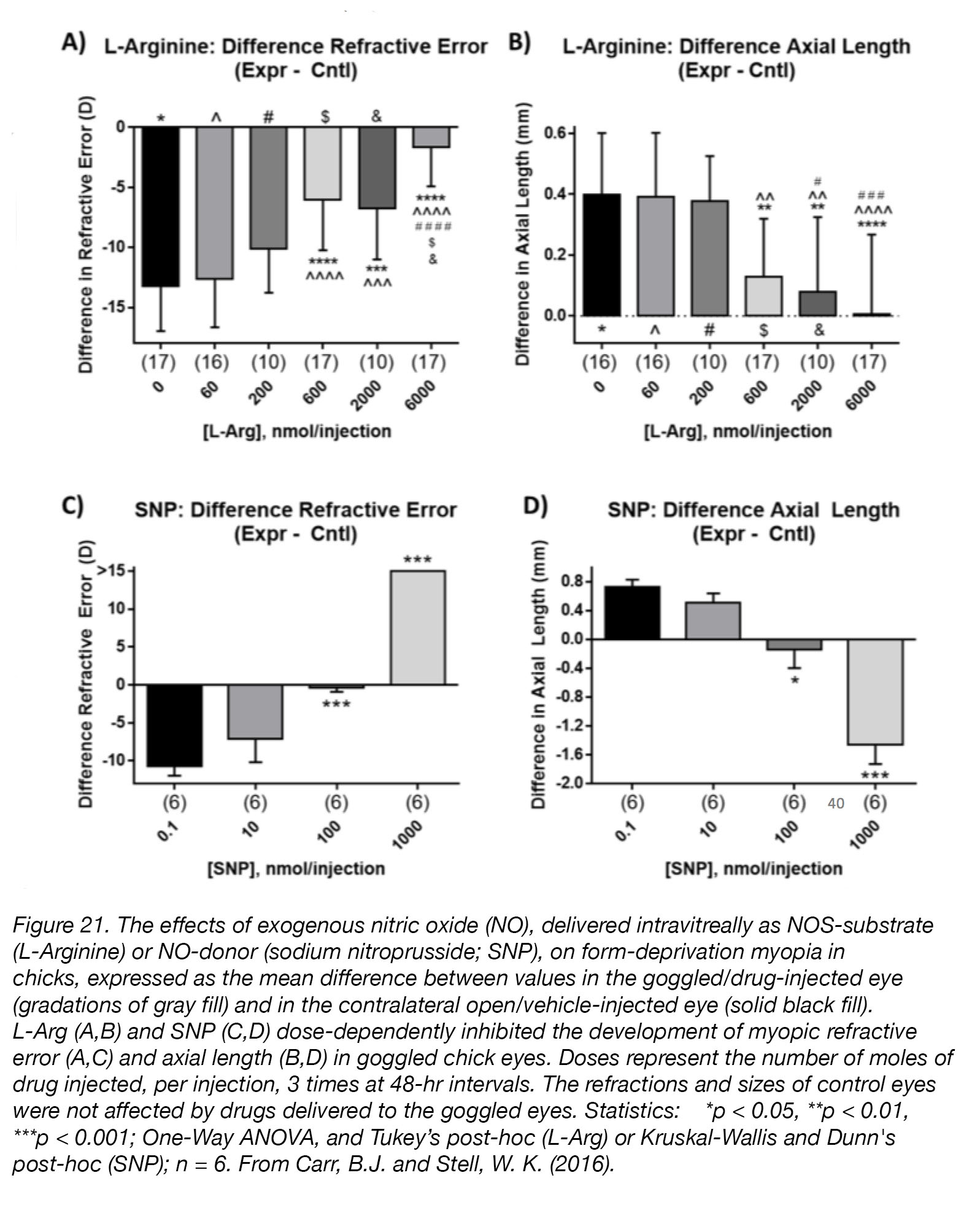
The investigation of the role of NO as a regulator of eye growth is still in its early stages, but preliminary results are promising. Non-specific inhibitors of NO synthesis, L-NAME [156] and L-NMMA [157], cause transient thinning of the choroid – a change correlated with myopiagenesis. Blockade of NO synthesis with L-NAME abolishes the rescue effect of unoccluded vision [158], and experiments in mice have verified that pharmacological blockade of NO synthesis increases myopia susceptibility [159]; this further implicates NO as a possible “stop” signal for eye growth. Finally, intravitreal injection of drugs that cause increased levels of NO in chick retina and RPE, dose-dependently inhibited the development of form-deprivation myopia (Fig. 21).
Figure 21. The effects of exogenous nitric oxide (NO), delivered intravitreally as NOS-substrate (L-Arginine; L-Arg) or NO-donor (sodium nitroprusside; SNP), on form-deprivation myopia in chicks, expressed as the mean difference between values in the goggled/drug-injected eye (gradations of gray fill) and in the contralateral open/vehicle-injected eye (solid black fill). L-Arg (A,B) and SNP (C,D) dose-dependently inhibited the development of myopic refractive error (A,C) and axial length (B,D) in goggled chick eyes. Doses represent the number of moles of drug injected, per injection, 3 times at 48-hr intervals. The refractions and sizes of control eyes were not affected by drugs delivered to the goggled eyes. Statistics: *p < 0.05, **p < 0.01, ***p < 0.001; One-Way ANOVA + Tukey’s post-hoc (L-Arg) or Kruskal-Wallis + Dunn’s post-hoc (SNP); n = 6. Reproduced with permission from Carr, B.J. and Stell, W. K. (2016) [48].
The mechanism of NO-mediated prevention of myopia remains to be elucidated. Increased retinal NO acts as a signal for light-adaptation [160-163], and retinal NO synthesis is increased under intense illumination [46]. NO donors in dark-adapted chicks mimic the adaptational effects of increased illumination [164], while NOS inhibitors mimic the effects of decreased illumination in light-adapted chicks [165]. Thus, increased NO may be a possible mechanism by which high-intensity environmental illumination protects against myopia. In addition, NO inhibits cell-cell coupling via gap junctions in the retina [166], and uncoupling of gap junctions containing connexin-35/36 (Cx35 in chicks) – which sharpens spatial tuning of retinal function – has also been implicated in myopia-inhibition [167] (Teves et al. [56].
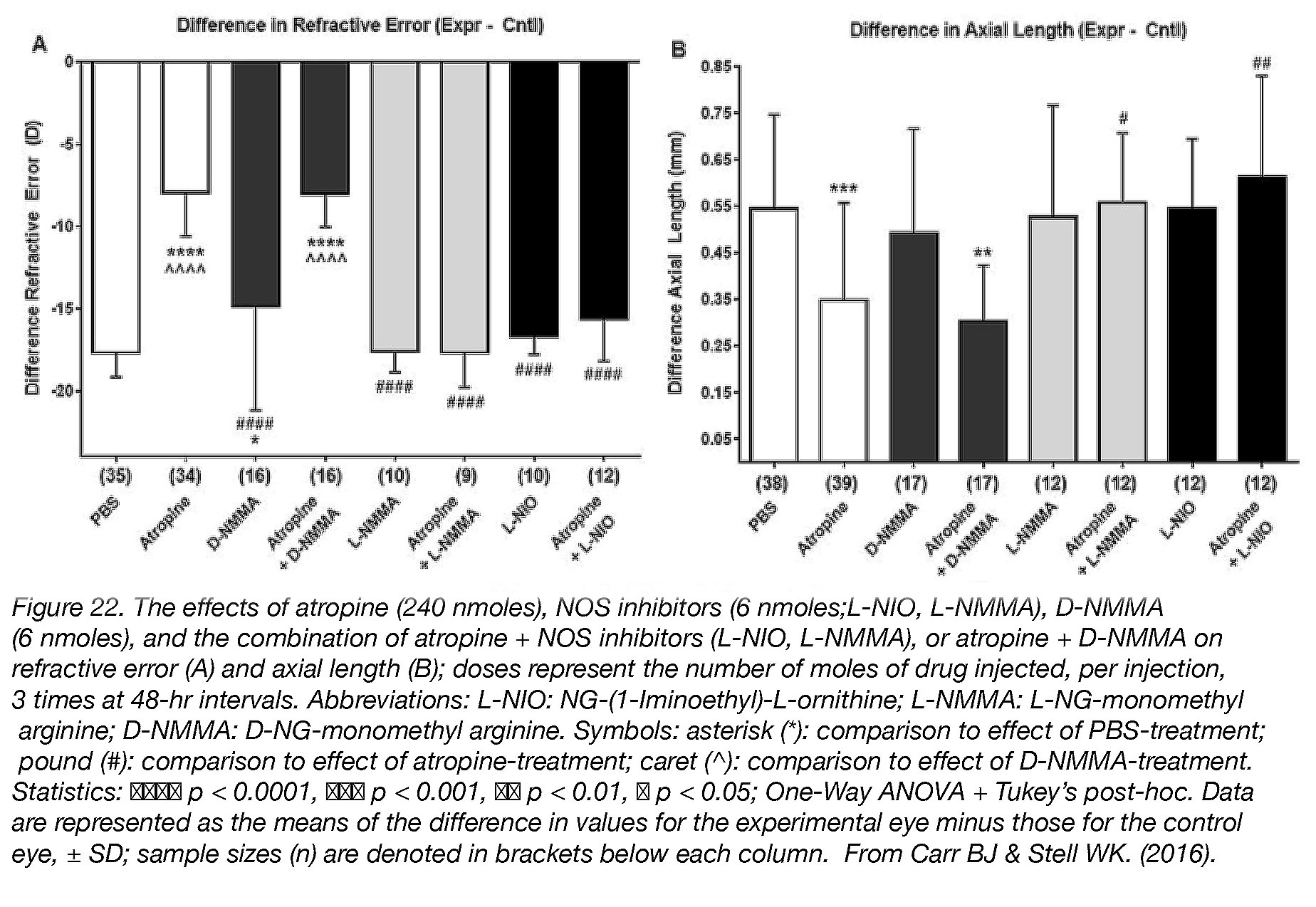
Although we do not know the receptors through which atropine may act, we do know that myopia-prevention by atropine is also dependent on induction of nitric oxide (NO). Simultaneous injection of atropine with nitric oxide synthase inhibitors – which blocks the ability of cells to make NO – completely abolishes all protective effects of atropine in chicks [48] (Fig. 22).
Figure 22. The effects of atropine (240 nmoles), NOS inhibitors (6 nmoles; L-NIO, L-NMMA), D-NMMA (6 nmoles), and the combination of atropine + NOS inhibitors (L-NIO, L-NMMA), or atropine + D-NMMA on refractive error (A) and axial length (B); doses represent the number of moles of drug injected, per injection, 3 times at 48-hr intervals. Abbreviations: L-NIO: NG-(1-Iminoethyl)-L-ornithine; L-NMMA: L-NG-monomethyl arginine; D-NMMA: D-NG-monomethyl arginine. Symbols: asterisk (*): comparison to effect of PBS-treatment; pound (#): comparison to effect of atropine-treatment; caret (^): comparison to effect of D-NMMA-treatment. Statistics: ●●●● p < 0.0001, ●●● p < 0.001, ●● p < 0.01, ● p < 0.05; One-Way ANOVA + Tukey’s post-hoc. Data are represented as the means of the differences in values for the experimental eye minus those for the control eye, ± SD; sample sizes (n) are denoted in brackets below each column. Reproduced with permission from Carr BJ & Stell WK. (2016) [9].
MANY OTHER MOLECULES CAN AFFECT EYE GROWTH
Adenosine Receptors and 7-methylzanthine (7-MX)
7-MX is a caffeine derivative, which has some affinity to adenosine receptors. Recent clinical trials of 7-MX have provided evidence that it may be useful in combatting axial myopia [168]. Children with a fast base-line rate of myopia progression, fed daily tablets containing 400mg 7-MX for >2 years, experienced slowing of axial elongation and myopia progression compared to placebo controls. This myopia-inhibiting effect halted as soon as 7-MX treatment was discontinued. The mechanism through which 7-MX may prevent myopia is currently unknown, but there are two working hypotheses: (i) that it causes stiffening of the sclera, which prevents axial elongation [169,170]; or (ii) that it blocks adenosine receptors in the retina and/or ciliary muscle, and consequently causes changes in acetylcholine and dopamine neurotransmitter release [168].
Alpha2-Adrenoceptors
Recent studies have shown that alpha2-adrenoceptor agonists can inhibit form-deprivation myopia in the chick [171]. Intravitreal injection of high concentration clonidine (200 nmoles) or guanfacine (20-200 nmoles) significantly inhibited changes in refractive error and axial elongation (Fig. 23). These studies are appealing, because the alpha2-adrenoceptor agonist brimonidine is already approved for use in humans, as a glaucoma treatment [172]. Thus, it would be much easier to translate these findings into a clinical trial than it would for experimental ligands such as mamba toxin-3. These data are very preliminary, but could yield new pharmaceutical targets for anti-myopia therapies lacking the negative side-effects of atropine treatment.
Figure 23. Effects of intravitreal injection of clonidine (top) and guanfacine (bottom) on FDM in chicks. 200 nmoles clonidine, and 20 and 200 nmoles guanfacine, significantly inhibited the induced difference in refractive error (a,c) and axial length (b,d), compared to PBS-controls. There was no significant effect of 2 or 20 nmoles clonidine-treatment or 2 nmoles guanfacine-treatment on difference in axial length. Statistics: ****p < 0.0001, ***p < 0.001, *p < 0.05; One-Way ANOVA + Tukey’s post-hoc. Data are represented as the means of the difference in values for the experimental eye minus those for the control eye, ± SD; sample sizes (n) are denoted in brackets below each column. Reproduced from Carr BJ. (2017). PhD Thesis. University of Calgary [123] and Carr BJ & Stell WK. [123,173].
Gamma-aminobutyric Acid (GABA)
Gamma (γ)-aminobutyric acid (GABA) is the primary mediator of inhibitory synaptic transmission in the central nervous system (CNS). For more detailed reviews of the localization and function of the GABA in the retina, please see previous Webvision chapters, “Neurotransmitters in the Retina” by Helga Kolb, and “GABAC Receptors in the Vertebrate Retina” by Haohua Qian.
There is significant evidence that GABA receptors are involved in the regulation of eye growth. Daily intravitreal injections of a wide variety of GABA receptor antagonists into form-deprived eyes resulted in reduction of eye growth (mostly in the equatorial dimension) [174]. One of the most effective anti-myopia GABAergic drugs is the GABAAOr/C antagonist (1,2,5,6-Tetrahydropyridin-4-yl)methylphosphinic acid (TPMPA), which has been shown to significantly inhibit goggle-induced myopic refractive error, vitreal chamber depth, and equatorial diameter in the chick [174] and guinea pig [175,176]. Novel GABAAOr/C drugs cis- and trans-3-ACPBPA also inhibit lens-induced myopia in chicks, the cis form being much more potent [177]. How GABAAOr/C antagonists may inhibit myopia is not known, and GABA can be expected to play a role in practically every visual processing mechanism in the retina; but there is some evidence of interaction with retinal dopaminergic systems, in the particular [178], as well as possible effects on scleral glycosaminoglycans [179].
Growth Factors: TGF-β and bFGF
Transforming growth factor-beta (TGF-β) and basic fibroblast growth factor (bFGF) seem to operate in a push-pull manner to regulate scleral growth during experimentally-induced myopia. Intravitreal injection of bFGF in chicks is protective against myopia, and co-administration of TGF-β with bFGF blocks this effect [180]. In the guinea pig, Chen et al. confirmed that bFGF prevents experimentally-induced myopia. They also reported that bFGF protein expression was decreased, while TGF-β expression was increased, in scleral tissues of eyes that had undergone light induced myopia for extended periods of time (15-30 days) [181]. The mechanism of bFGF-mediated myopia inhibition has not been elucidated. Attempts were made to link bFGF with the dopaminergic system, because bFGF protein content is upregulated during light and it is co-localized with tyrosine-hydroxylase – the cell marker for dopaminergic amacrine cells [180]. However, other studies revealed that the two pathways are likely not related [88].
Peptide Hormones: Glucagon and Insulin
Glucagon is a pancreatic peptide hormone whose best-known role in mammals is to raise the concentration of glucose in the blood; insulin, another pancreatic peptide hormone, plays the opposite role – to lower blood sugar levels. However, many (perhaps all) gut peptides and their receptors are also expressed by neurons in the central and peripheral nervous systems, where they presumably play neuromodulatory roles. Glucagon-positive cells in the retina consist of a subset of amacrine cells in the inner retina of birds and reptiles, about 40% of which are also GABAergic [182,183]. The activity of glucagon amacrine cells in the chick retina changes dramatically in response to defocus, as revealed by changes in these cells’ content of an activity-inducible transcription factor, early growth response protein 1 (EGR-1 or ZENK). Positive-lens defocus, or removal of the diffuser after inducing form-deprivation myopia, causes stronger and more widespread labeling of EGR-1, whereas negative-lens defocus or form-deprivation results in little or no labeling [184]. The activity-dependent induction of Egr-1 is best interpreted as an indicator of cell depolarization, and therefore is an indicator of increased transmitter release. Egr-1 is likely to upregulate the transcription of genes whose products are important in functional compensation for prolonged increases in cell activity, rather than directly to affect downstream actions of the activated cell such as those involved in myopia-inhibition.
In the chick, injection of a glucagon agonist into form-deprived eyes inhibits myopia development [185], and injection of a glucagon antagonist blocks myopia recovery from clear vision [186]. Intravitreal injection of insulin has the opposite effect, acting as a powerful myopiagenic agent [187]. Glucagon inhibits the development of myopia in chicks, even after most amacrine cells have been ablated by neurotoxins, suggesting that (in chicks at least) glucagon amacrine cells could be the final or near-final output of “stop” signals acting on the RPE (Beloukhina N, et al. IOVS 2005; 46: ARVO E-Abstract 3337).
The relevance of these interesting findings in the chick model to the control of refractive development in mammalian eyes remains uncertain. A comparable role of glucagon in the retina of mammals remains elusive. To date, glucagon has not been identified or localized in any mammalian retina; and although many other members of the glucagon polypeptide family are present, their expression is not changed upon treatment of the eyes with form-deprivation stimuli. However, other kinds of amacrine cells, which have been labelled by similar focus- and defocus-dependent induction of Egr-1 in macaque monkey [188,189] and tree shrew (Stell WK, et al. IOVS 2004; 45: ARVO E-Abstract 1159) are plausible candidates for playing similar roles in the control of eye growth.
Retinoic Acid
Retinoic acid (RA) is member of the retinoid family of compounds, which also includes Vitamin A. Retinoic acid stands out among other candidate regulators in the control of eye growth, because its levels are bi-directionally modulated in response to opposing defocus stimuli [190-193]. In the chick, retinal and choroidal RA levels and mRNA expression of its synthesizing enzyme, aldehyde dehydrogenase-2 (AHD2), are predictably changed in response to defocus stimuli; increasing in response to negative defocus and decreasing in response to positive defocus [192-194]. Treatment of form-deprived chicks with a blocker of RA synthesis, disulfiram, caused myopia inhibition, but this effect was not carried over in response to lens-induced myopia [194]. RA might act as a regulator of eye growth by inducing changes in scleral extracellular matrix [195] or modulating cell-cell coupling [196,197].
CHOROIDAL CHANGES CORRESPOND TO CHANGES IN EYE GROWTH
Although the origin of homeostatic growth-regulating signals is most likely the retina, experimental and therapeutic treatments may affect any target within other ocular tissues – the retinal pigment epithelium (RPE), choroid, and/or sclera most likely. It is unlikely that a single kind of molecule is responsible for transmission of growth-regulating signals, because of difficulties in crossing the occluding junctions of the RPE (a major component of the blood-retina-barrier), and drug loss due to protein binding and dilution of the drug during en route transport into cells. More likely, a cascade of molecules may relay signals from retina-to-RPE, RPE-to-choroid, and then choroid-to-sclera (Fig. 16).
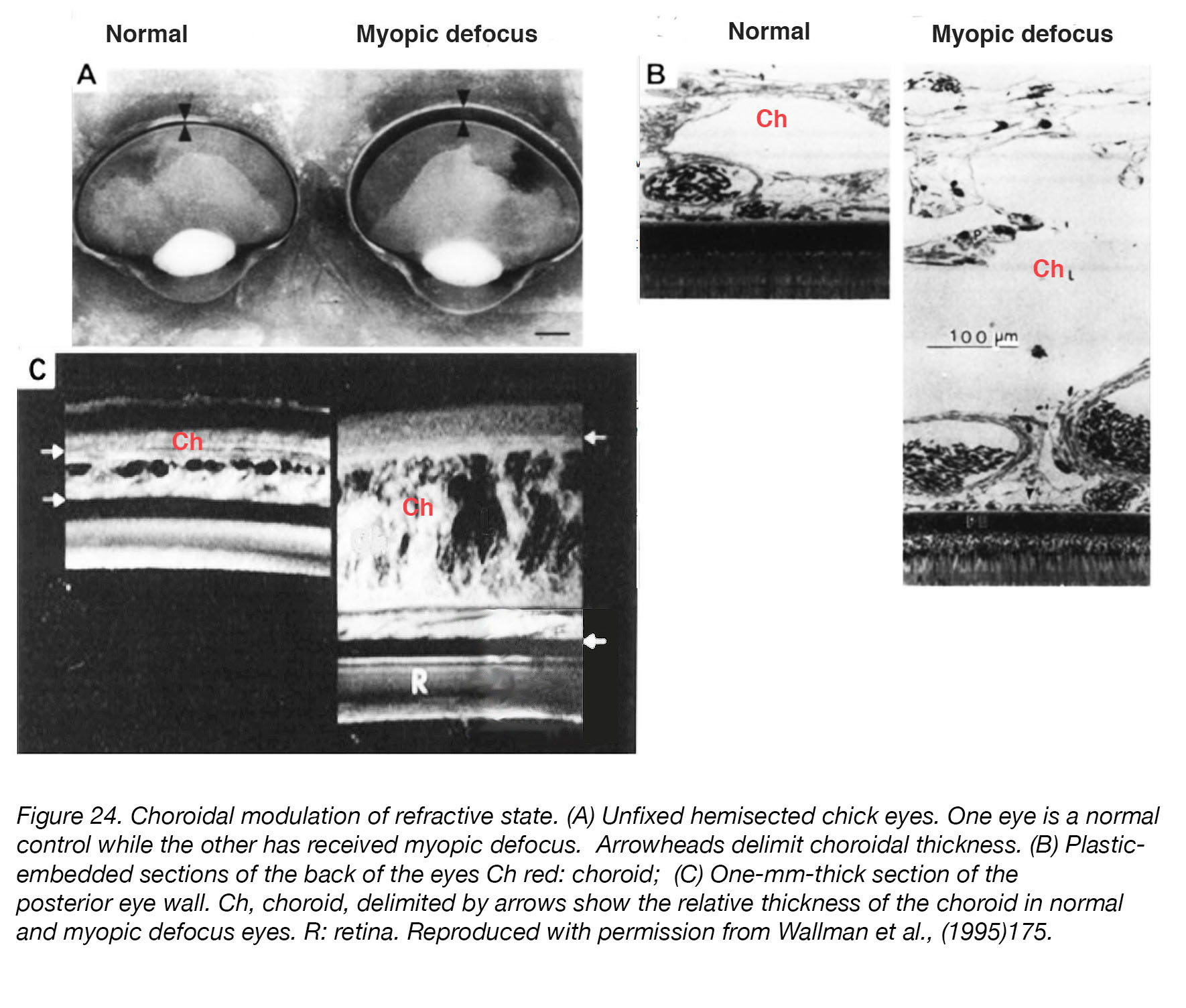
Interestingly, the choroid responds to induced defocus in a compensatory fashion [198] – thickening in response to myopic defocus (positive lenses, removal of myopia-inducing goggles) and myopia-inhibiting treatments [121,156-158,199], and thinning in response to diffusers or hyperopic defocus (negative lenses) (Fig. 24). Whether the directional changes in the choroid are responsible for scleral changes in eye growth – or just an initial compensatory step/by-product of other mechanisms that are themselves the primary regulators – remains to be determined, although the authors of a recent study have reported that changes in choroidal thickness are not predictive of changes in eye growth in eyes with experimentally-induced myopia200. Nevertheless, it may be assumed that the choroid should play some role in the regulation of ocular refraction and/or the cause(s) and prevention of myopia [201], and that it may be the source of growth-regulating signals that act on the sclera [202].
Figure 24. Choroidal modulation of refractive state. (A) Unfixed hemisected chick eyes. One eye is a normal control while the other has received myopic defocus. Arrowheads delimit choroidal thickness. (B) Plastic-embedded sections of the back of the eyes. Ch (red): choroid; R: retina. (C) One-mm-thick section of the posterior eye wall. Ch: choroid, delimited by arrows show the relative thickness of the choroid in normal and myopic defocus eyes. Reproduced with permission from Wallman et al., (1995) [198].
Human Therapies and Recommendations for Myopia Prevention
Pharmaceutical Interventions: Atropine
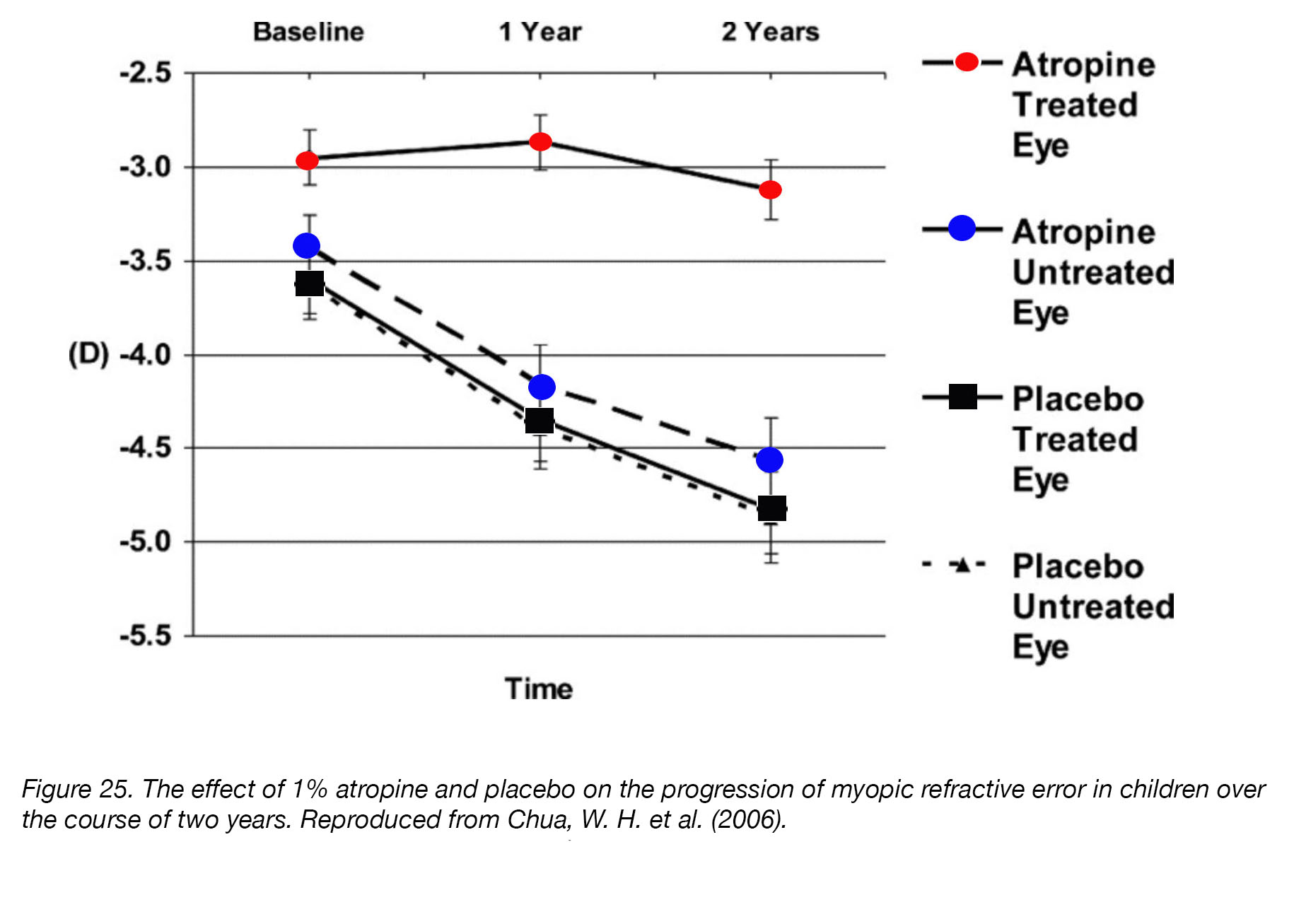
Although many neurotransmitter/neuromodulator agonists and antagonists have been investigated for their ability to control eye growth, very few laboratory discoveries have been translated into clinical therapies. The most widely used pharmaceutical agent, atropine, was championed almost 40 years ago by Bedrossian [203] and subsequently became the subject of numerous clinical trials and scientific scrutiny. In 2006, Chua et al. published the results of the Atropine in the Treatment Of Myopia (ATOM) study [96]. This was a two-year, comprehensive, randomized, placebo-controlled, double-blind trial of 346 Asian children to determine the effectiveness and safety of daily 1% atropine eye drops for the prevention of myopia progression. ATOM was significant in that it was one of the first clinical trials with double-blind and placebo-controlled groups, which provided a much more objective measurement of the effects of atropine for human myopia. The outcomes revealed that atropine significantly reduced the mean progression of myopic refractive error: 84% of placebo-treated children experienced myopic progression of -0.5 D or more, while only 34% of atropine-treated children experienced the same (Fig. 25). No serious adverse effects were reported, but the common side-effects – photophobia, glare, allergic reaction, and headaches caused by blurred vision – caused some children to drop out. In addition, a subset of children experienced a “rebound effect”, in which myopia returned and progressed much more rapidly after atropine-treatment had ceased [204]. Chia et al. [95] repeated the ATOM study (ATOM2), using lower concentrations of atropine (0.5%, 0.1%, and 0.01%) for two years, and found that the drug retained most of its efficacy at these lower concentrations. Minimal side effects were reported, but many children continued to suffer from blurred vision, conjunctivitis, and eyelid dermatitis at the higher concentrations. The “rebound effect” was also still present in the higher-concentration treatment groups (0.5% and 0.1%) [205].
Figure 25. The effect of 1% atropine and placebo on the progression of myopic refractive error in children over the course of two years. Reproduced from Chua, W. H. et al. (2006) [96].
In an attempt to address the rebound effect, the study was repeated, but with a different treatment schedule [206]. Children were treated with 0.5%, 0.1%, or 0.01% atropine for two years, and then treatment was discontinued for one year to see whether rebound would occur. Treatment was restarted only in children who experienced myopic progression of -0.50 diopters or more in at least one eye, and then continued for another two years. It was found that there was an inverse dose-related increase in myopia by the end of phase 2 (period with no treatment – ‘wash-out’) caused by rebound, resulting in the greatest effect of the 0.01% treatment group. Children requiring a second round of atropine-treatment were 24%, 59%, and 68% for the 0.01%, 0.1%, and 0.5% groups, respectively. Results from the second round of treatment confirmed that the most effective concentration of atropine was 0.01%; children treated with 0.01% atropine had the least rebound during the wash-out period, but still experienced significant inhibition of myopia-progression during treatment (Fig. 26). Side-effects for the 0.01%-treatment group were mild – only minimal pupillary dilation, minimal loss of accommodation, and no significant loss of near-vision – compared with those at the higher concentrations.
Figure 26. Mean change in refractive error over time within different treatment groups (atropine 0.01%, 0.1%, and 0.5%). Error bars represent 1 standard deviation. Reproduced from Chia, A., Lu, Q. S. & Tan, D. (2016) [206].
Corneal-Reshaping Lenses: Orthokeratology
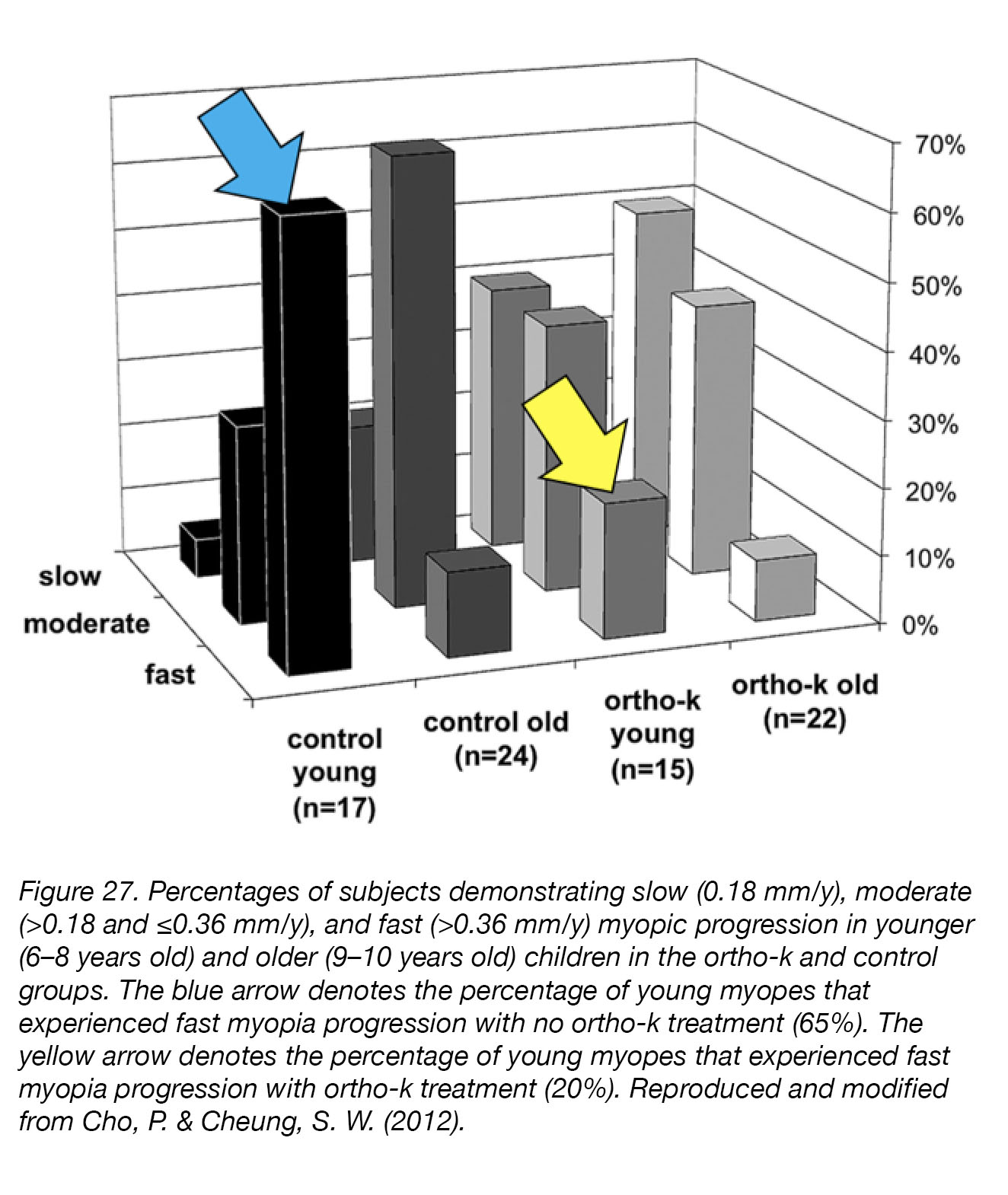
Orthokeratology (Ortho-K, Overnight Vision Correction, Corneal Refractive Therapy) uses gas-permeable contact lenses, worn and night and removed during the day, to temporarily reduce refractive errors. The lenses reshape the cornea while the user is sleeping, are removed in the morning, and refractive correction is maintained well throughout the day. Not only does Ortho-K correct myopia without drugs or surgery, but clinical trials of Ortho-K have demonstrated a strong correlation between Orth-K treatment and inhibition of myopia progression [207]. The Longitudinal Orthokeratology Research in Children (LORIC) study, in Hong Kong, was a non-randomized two-year pilot study that investigated the ability of overnight Ortho-K lens wear (i) to correct myopic vision, and (ii) to slow the progression of myopia (n=35) [208]. Positive results from this small pilot study led to the first larger-scale, single-masked, randomized clinical trial named the Retardation Of Myopia In Orthokeratology (ROMIO) study [209], in which 78 children (37 Ortho-K, 41 controls) took part for two years. The children wearing Ortho-K lenses experienced significantly slower axial elongation during the study than those wearing single-vision lenses, and at the end of the treatment period had significantly shorter eyes (Fig. 27). Some children (5) did drop out of the study for ocular health reasons related to lens-wear, but their vision was not permanently affected once Ortho-K treatment was stopped. The mechanism by which orthokeratology inhibited myopia progression remains unclear, but studies have suggested that Ortho-K lenses may induce relative peripheral myopia, as well as increases in spherical aberration and amplitude of accommodation210. However, alternative mechanisms – such as neurally mediated reflex control of ocular elongation or corneal curvature – have not yet been ruled out.
Figure 27. Percentages of subjects demonstrating slow (0.18 mm/y), moderate (>0.18 and ≤0.36 mm/y), and fast (>0.36 mm/y) myopia-progression in younger (6–8 years old) and older (9–10 years old) children, in the Ortho-K and control groups. The blue arrow indicates the percentage of young myopes that experienced rapid myopia-progression without Ortho-K treatment (65%), while the yellow arrow indicates the percentage of young myopes that experienced rapid myopia progression with Ortho-K treatment (20%). Reproduced and modified from Cho, P. & Cheung, S. W. (2012) [209].
Alternative Strategies: High Environmental Illuminance
Human trials investigating the effects of high-intensity outdoor light on myopia prevention have so far been very promising [37,39,40] (Fig. 10). Sending children outdoors is by far the least invasive treatment-option currently available. In countries where the intensity of outdoor light is generally lower, because of air pollution or short duration of natural daylight – such as Canada or Scandinavia in the winter, or Beijing year-around – sunlight therapy could be supplemented in the form of SAD lights (approved and used for Seasonal Affective Disorder), which have an output of ~2,000-10,000 lux and are sold to combat depressive symptoms brought on by seasonal affective disorder. This alternative will become even more attractive, when the spectral region responsible for myopia-prevention in primates has been identified.
WHERE DO WE GO FROM HERE?
The world is experiencing a “myopia boom” [2], and viable treatment options are becoming more crucial than ever before. Atropine is considered to be the best pharmaceutical treatment we have. It is the only one currently available, and is rather widely accepted and used, despite remaining off-label for the purpose of treating myopia. Advocates of atropine use would claim that the occurrence of unwanted side-effects and its unknown mechanism of action are not serious enough drawbacks to warrant discontinuation of therapy. Unlike atropine, single-vision lenses do not prevent continued myopia progression, which can lead to blindness-inducing vision problems later in life; and overall, topical atropine-treatment in children has been proven to be effective and quite safe. Parents should be aware of the most common side effects: possible photophobia due to pupil dilation, impairment of near-vision due to paralysis of the ciliary muscle, allergic reactions, and rebound after cessation of treatment. It should also be noted, that not all children will respond to atropine-treatment [211].
It is imperative to determine the mechanism through which atropine inhibits myopia, because a better agent might be found. Ideally, an agent as effective topically as atropine, but completely lacking the unacceptable side-effects of even 0.01% atropine eye-drops – mild photophobia, blur, and allergies. There is evidence that mAChRs may not be the receptors that mediate myopia-inhibition [114], and learning the true targets could effectively eliminate atropine’s three most common side-effects. Lowering the required concentrations or treatment frequency could mitigate the allergic reactions caused by daily atropine use, and using a non-mAChR-specific drug would eliminate both photophobia and loss of accommodation, which are mediated by known target receptors of atropine (mAChRs).
Retrospective reviews have been performed to determine the safety and efficacy of Ortho-K lens-wear; however, although gas-permeable Ortho-K lenses have been determined to be quite safe [207,212], caution is still advised. Long-term use of these lenses may be required, which can increase the risk of microbial keratitis, and discontinuation of use has resulted in a rebound effect similar to that seen in pharmacological treatments of myopia [204,205,210]. Recently, a study was performed that combined the anti-myopia effects of atropine (0.01%) and Ortho-K. The authors reported that combining the two therapies resulted in a significantly greater effect than Ortho-K treatment alone [213]. Long-term studies on the safety and efficacy of cornea-reshaping lenses are vital; whether overall eye health can be maintained while using lenses that change eye shape by applying a significant amount of physical pressure to corneal cells for many hours every day remains to be determined.
Outdoor-light therapy may offer the ideal treatment for myopia. Not only does encouraging children to play outside and combat other major health concerns – such as childhood obesity, juvenile diabetes, and depression – but also, light therapy presents little to no serious health concerns or side-effects compared to those of other available myopia-treatments. Exposure to potentially-harmful UV radiation can be avoided by wearing UVA/UVB-blocking sunglasses and sunscreen. Alternative light sources such as commercially available SAD lights do not contain UV-radiation, and have been shown to be nearly equally effective in myopia-prevention in animal models. This approach seems to be particularly promising, if further testing of non-human primates, followed by clinical trials, confirms that long (rather than short) wavelengths of visible light are the ones that can protect humans against myopia, as blue-light can be potentially harmful to the retina [214].
Although we have made significant progress in determining possible treatments for myopia, much still needs to be done before we understand fully the homeostatic mechanisms responsible for regulation of eye growth. New discoveries about the way visual feedback regulates eye size may lead to new treatment options for myopia, and further our understanding of a fundamental mechanism of human eye development and emmetropization, a poorly understood, but highly important function of visual processing in the retina.
REFERENCES
1 Foster, P. J. & Jiang, Y. Epidemiology of myopia. Eye (Lond) 28, 202-208, doi:10.1038/eye.2013.280 (2014). [PubMed]
2 Dolgin, E. The myopia boom. Nature 519, 276-278, doi:10.1038/519276a (2015). [PubMed]
3 The Impact of Myopia and High Myopia. World Health Organization, doi: http://www.who.int/blindness/causes/MyopiaReportforWeb.pdf (2015).
4 Wong, T. Y., Ferreira, A., Hughes, R., Carter, G. & Mitchell, P. Epidemiology and disease burden of pathologic myopia and myopic choroidal neovascularization: an evidence-based systematic review. Am J Ophthalmol 157, 9-25.e12, doi:10.1016/j.ajo.2013.08.010 (2014). [PubMed]
5 Prevalence of myopia and hyperopia in 6- to 72-month-old african american and Hispanic children: the multi-ethnic pediatric eye disease study. Ophthalmology 117, 140-147.e143, doi:10.1016/j.ophtha.2009.06.009 (2010). [PubMed]
6 Wen, G. et al. Prevalence of myopia, hyperopia, and astigmatism in non-Hispanic white and Asian children: multi-ethnic pediatric eye disease study. Ophthalmology 120, 2109-2116, doi:10.1016/j.ophtha.2013.06.039 (2013). [PubMed]
7 French, A. N., Morgan, I. G., Burlutsky, G., Mitchell, P. & Rose, K. A. Prevalence and 5- to 6-year incidence and progression of myopia and hyperopia in Australian schoolchildren. Ophthalmology 120, 1482-1491, doi:10.1016/j.ophtha.2012.12.018 (2013). [PubMed]
8 Verhoeven, V. J. et al. Genome-wide meta-analyses of multiancestry cohorts identify multiple new susceptibility loci for refractive error and myopia. Nat Genet 45, 314-318, doi:10.1038/ng.2554 (2013). [PubMed]
9 Dirani, M. et al. Refractive errors in twin studies. Twin Res Hum Genet 9, 566-572, doi:10.1375/183242706778024955 (2006). [PubMed]
10 Ding, X. et al. Distribution and heritability of peripheral eye length in Chinese children and adolescents: the Guangzhou Twin Eye Study. Invest Ophthalmol Vis Sci 54, 1048-1053, doi:10.1167/iovs.12-10066 (2013). [PubMed]
11 Kim, M. H. et al. Heritability of myopia and ocular biometrics in Koreans: the healthy twin study. Invest Ophthalmol Vis Sci 54, 3644-3649, doi:10.1167/iovs.12-11254 (2013). [PubMed]
12 Mutti, D. O., Mitchell, G. L., Moeschberger, M. L., Jones, L. A. & Zadnik, K. Parental myopia, near work, school achievement, and children’s refractive error. Invest Ophthalmol Vis Sci 43, 3633-3640 (2002). [PubMed]
13 Ip, J. M. et al. Ethnic differences in the impact of parental myopia: findings from a population-based study of 12-year-old Australian children. Invest Ophthalmol Vis Sci 48, 2520-2528, doi:10.1167/iovs.06-0716 (2007). [PubMed]
14 Lam, D. S. et al. The effect of parental history of myopia on children’s eye size and growth: results of a longitudinal study. Invest Ophthalmol Vis Sci 49, 873-876, doi:10.1167/iovs.06-1097 (2008). [PubMed]
15 Xiang, F., He, M. & Morgan, I. G. The impact of parental myopia on myopia in Chinese children: population-based evidence. Optom Vis Sci 89, 1487-1496, doi:10.1097/OPX.0b013e31826912e0 (2012). [PubMed]
16 Guo, L. et al. Prevalence and associated factors of myopia among primary and middle school-aged students: a school-based study in Guangzhou. Eye (Lond), doi:10.1038/eye.2016.39 (2016). [PubMed]
17 Rose, K. A., Morgan, I. G., Smith, W. & Mitchell, P. High heritability of myopia does not preclude rapid changes in prevalence. Clin Experiment Ophthalmol 30, 168-172, doi:10.1046/j.1442-9071.2002.00521.x (2002). [PubMed]
18 Ramessur, R., Williams, K. M. & Hammond, C. J. Risk factors for myopia in a discordant monozygotic twin study. Ophthalmic Physiol Opt 35, 643-651, doi:10.1111/opo.12246 (2015). [PubMed]
19 Wallman, J. & Winawer, J. Homeostasis of eye growth and the question of myopia. Neuron 43, 447-468, doi:10.1016/j.neuron.2004.08.008 (2004). [PubMed]
20 Hubel, D. H., Wiesel, T. N. & LeVay, S. Functional architecture of area 17 in normal and monocularly deprived macaque monkeys. Cold Spring Harb Symp Quant Biol 40, 581-589, doi:10.1101/SQB.1976.040.01.054 (1976). [PubMed]
21 Wiesel, T. N. & Raviola, E. Myopia and eye enlargement after neonatal lid fusion in monkeys. Nature 266, 66-68, doi:10.1038/266066a0 (1977). [PubMed]
22 Norton, T. T., Casagrande, V. A. & Sherman, S. M. Loss of Y-cells in the lateral geniculate nucleus of monocularly deprived tree shrews. Science 197, 784-786 (1977). [PubMed]
23 Wallman, J., Turkel, J. & Trachtman, J. Extreme myopia produced by modest change in early visual experience. Science 201, 1249-1251, doi:10.1126/science.694514 (1978). [PubMed]
24 Schaeffel, F., Glasser, A. & Howland, H. C. Accommodation, refractive error and eye growth in chickens. Vision Res 28, 639-657, doi:10.1016/0042-6989(88)90113-7 (1988). [PubMed]
25 Schaeffel, F. & Feldkaemper, M. Animal models in myopia research. Clin Exp Optom 98, 507-517, doi:10.1111/cxo.12312 (2015). [PubMed]
26 Troilo, D., Gottlieb, M. D. & Wallman, J. Visual deprivation causes myopia in chicks with optic nerve section. Curr Eye Res 6, 993-999, doi:10.3109/02713688709034870 (1987). [PubMed]
27 Wallman, J., Gottlieb, M. D., Rajaram, V. & Fugate-Wentzek, L. A. Local retinal regions control local eye growth and myopia. Science 237, 73-77, doi:10.1126/science.3603011 (1987). [PubMed]
28 Napper, G. A. et al. The duration of normal visual exposure necessary to prevent form deprivation myopia in chicks. Vision Res 35, 1337-1344, doi:10.1016/0042-6989(94)00226-C (1995). [PubMed]
29 Zhu, X., Winawer, J. A. & Wallman, J. Potency of myopic defocus in spectacle lens compensation. Invest Ophthalmol Vis Sci 44, 2818-2827, doi:10.1167/iovs.02-0606 (2003). [PubMed]
30 Gwiazda, J. et al. A randomized clinical trial of progressive addition lenses versus single vision lenses on the progression of myopia in children. Invest Ophthalmol Vis Sci 44, 1492-1500 (2003). [PubMed]
31 Berntsen, D. A., Sinnott, L. T., Mutti, D. O. & Zadnik, K. A randomized trial using progressive addition lenses to evaluate theories of myopia progression in children with a high lag of accommodation. Invest Ophthalmol Vis Sci 53, 640-649, doi:10.1167/iovs.11-7769 (2012). [PubMed]
32 Galvis, V. et al. Topical Atropine in the Control of Myopia. Med Hypothesis Discov Innov Ophthalmol 5, 78-88 (2016). [PubMed]
33 Chung, K., Mohidin, N. & O’Leary, D. J. Undercorrection of myopia enhances rather than inhibits myopia progression. Vision Res 42, 2555-2559 (2002). [PubMed]
34 Adler, D. & Millodot, M. The possible effect of undercorrection on myopic progression in children. Clin Exp Optom 89, 315-321, doi:10.1111/j.1444-0938.2006.00055.x (2006). [PubMed]
35 Sun, Y. Y. et al. Effect of uncorrection versus full correction on myopia progression in 12-year-old children. Graefes Arch Clin Exp Ophthalmol 255, 189-195, doi:10.1007/s00417-016-3529-1 (2017). [PubMed]
36 French, A. N., Ashby, R. S., Morgan, I. G. & Rose, K. A. Time outdoors and the prevention of myopia. Exp Eye Res 114, 58-68, doi:10.1016/j.exer.2013.04.018 (2013). [PubMed]
37 Read, S. A., Collins, M. J. & Vincent, S. J. Light Exposure and Eye Growth in Childhood. Invest Ophthalmol Vis Sci 56, 6779-6787, doi:10.1167/iovs.14-15978 (2015). [PubMed]
38 Rucker, F. J. The role of luminance and chromatic cues in emmetropisation. Ophthalmic Physiol Opt 33, 196-214, doi:10.1111/opo.12050 (2013). [PubMed]
39 Rose, K. A. et al. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology 115, 1279-1285, doi:10.1016/j.ophtha.2007.12.019 (2008). [PubMed]
40 He, M. et al. Effect of Time Spent Outdoors at School on the Development of Myopia Among Children in China: A Randomized Clinical Trial. Jama 314, 1142-1148, doi:10.1001/jama.2015.10803 (2015). [PubMed]
41 Smith, E. L., 3rd, Hung, L. F. & Huang, J. Protective effects of high ambient lighting on the development of form-deprivation myopia in rhesus monkeys. Invest Ophthalmol Vis Sci 53, 421-428, doi:10.1167/iovs.11-8652 (2012). [PubMed]
42 Smith, E. L., 3rd, Hung, L. F., Arumugam, B. & Huang, J. Negative lens-induced myopia in infant monkeys: effects of high ambient lighting. Invest Ophthalmol Vis Sci 54, 2959-2969, doi:10.1167/iovs.13-11713 (2013). [PubMed]
43 Ashby, R., Ohlendorf, A. & Schaeffel, F. The effect of ambient illuminance on the development of deprivation myopia in chicks. Invest Ophthalmol Vis Sci 50, 5348-5354, doi:10.1167/iovs.09-3419 (2009). [PubMed]
44 Ashby, R. S. & Schaeffel, F. The effect of bright light on lens compensation in chicks. Invest Ophthalmol Vis Sci 51, 5247-5253, doi:10.1167/iovs.09-4689 (2010). [PubMed]
45 Ashby, R. Animal Studies and the Mechanism of Myopia-Protection by Light? Optom Vis Sci 93, 1052-1054, doi:10.1097/opx.0000000000000978 (2016). [PubMed]
46 Hoshi, H., Sato, M., Oguri, M. & Ohtsuka, T. In vivo nitric oxide concentration in the vitreous of rat eye. Neurosci Lett 347, 187-190, doi:10.1016/s0304-3940(03)00717-1 (2003). [PubMed]
47 Feldkaemper, M. & Schaeffel, F. An updated view on the role of dopamine in myopia. Exp Eye Res 114, 106-119, doi:10.1016/j.exer.2013.02.007 (2013). [PubMed]
48 Carr, B. J. & Stell, W. K. Nitric Oxide (NO) Mediates the Inhibition of Form-Deprivation Myopia by Atropine in Chicks. Scientific Reports 6, 9, doi:10.1038/s41598-016-0002-7 (2016). [PubMed]
49 Nickla, D. L., Lee, L. & Totonelly, K. Nitric oxide synthase inhibitors prevent the growth-inhibiting effects of quinpirole. Optom Vis Sci 90, 1167-1175, doi:10.1097/opx.0000000000000041 (2013). [PubMed]
50 Zhu, X., Park, T. W., Winawer, J. & Wallman, J. In a matter of minutes, the eye can know which way to grow. Invest Ophthalmol Vis Sci 46, 2238-2241, doi:10.1167/iovs.04-0956 (2005). [PubMed]
51 Rucker, F. J. & Wallman, J. Chick eyes compensate for chromatic simulations of hyperopic and myopic defocus: evidence that the eye uses longitudinal chromatic aberration to guide eye-growth. Vision Res 49, 1775-1783, doi:10.1016/j.visres.2009.04.014 (2009). [PubMed]
52 Rucker, F. J. & Wallman, J. Cone signals for spectacle-lens compensation: differential responses to short and long wavelengths. Vision Res 48, 1980-1991, doi:10.1016/j.visres.2008.06.003 (2008). [PubMed]
53 Rohrer, B., Schaeffel, F. & Zrenner, E. Longitudinal chromatic aberration and emmetropization: results from the chicken eye. J Physiol 449, 363-376 (1992). [PubMed]
54 Wildsoet, C. F., Howland, H. C., Falconer, S. & Dick, K. Chromatic aberration and accommodation: their role in emmetropization in the chick. Vision Res 33, 1593-1603 (1993). [PubMed]
55 Foulds, W. S., Barathi, V. A. & Luu, C. D. Progressive myopia or hyperopia can be induced in chicks and reversed by manipulation of the chromaticity of ambient light. Invest Ophthalmol Vis Sci 54, 8004-8012, doi:10.1167/iovs.13-12476 (2013). [PubMed]
56 Benavente-Perez, A. et al. Abstracts from the 15th International Myopia Conference. Eye and Vision 3, 1-11, doi:10.1186/s40662-016-0057-3 (2016). [PubMed]
57 Jiang, L. et al. Interactions of chromatic and lens-induced defocus during visual control of eye growth in guinea pigs (Cavia porcellus). Vision Res 94, 24-32, doi:10.1016/j.visres.2013.10.020 (2014). [PubMed]
58 Qian, Y. F. et al. Transfer from blue light or green light to white light partially reverses changes in ocular refraction and anatomy of developing guinea pigs. J Vis 13, doi:10.1167/13.11.16 (2013). [PubMed]
59 Long, Q., Chen, D. & Chu, R. Illumination with monochromatic long-wavelength light promotes myopic shift and ocular elongation in newborn pigmented guinea pigs. Cutan Ocul Toxicol 28, 176-180, doi:10.3109/15569520903178364 (2009). [PubMed]
60 Smith, E. L., 3rd et al. Effects of Long-Wavelength Lighting on Refractive Development in Infant Rhesus Monkeys. Invest Ophthalmol Vis Sci 56, 6490-6500, doi:10.1167/iovs.15-17025 (2015). [PubMed]
61 Charman, W. N. Keeping the world in focus: how might this be achieved? Optom Vis Sci 88, 373-376, doi:10.1097/OPX.0b013e31820b052b (2011). [PubMed]
62 Morgan, I. G., Ashby, R. S. & Nickla, D. L. Form deprivation and lens-induced myopia: are they different? Ophthalmic Physiol Opt 33, 355-361, doi:10.1111/opo.12059 (2013). [PubMed]
63 Fischer, A. J., Morgan, I. G. & Stell, W. K. Colchicine causes excessive ocular growth and myopia in chicks. Vision Res 39, 685-697, doi:10.1016/S0042-6989(98)00178-3 (1999). [PubMed]
64 Ehrlich, D., Sattayasai, J., Zappia, J. & Barrington, M. Effects of selective neurotoxins on eye growth in the young chick. Ciba Found Symp 155, 63-84; discussion 84-68 (1990). [PubMed]
65 Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat Methods 11, 783-784, doi:10.1038/nmeth.3047 (2014). [PubMed]
66 Oishi, I., Yoshii, K., Miyahara, D., Kagami, H. & Tagami, T. Targeted mutagenesis in chicken using CRISPR/Cas9 system. Sci Rep 6, 23980, doi:10.1038/srep23980 (2016). [PubMed]
67 Chakraborty, R. & Pardue, M. T. Molecular and Biochemical Aspects of the Retina on Refraction. Prog Mol Biol Transl Sci 134, 249-267, doi:10.1016/bs.pmbts.2015.06.013 (2015). [PubMed]
68 Park, H. et al. Retinal degeneration increases susceptibility to myopia in mice. Mol Vis 19, 2068-2079 (2013). [PubMed]
69 Park, H. et al. Visually-driven ocular growth in mice requires functional rod photoreceptors. Invest Ophthalmol Vis Sci 55, 6272-6279, doi:10.1167/iovs.14-14648 (2014). [PubMed]
70 Pardue, M. T. et al. High susceptibility to experimental myopia in a mouse model with a retinal on pathway defect. Invest Ophthalmol Vis Sci 49, 706-712, doi:10.1167/iovs.07-0643 (2008). [PubMed]
71 Fujikado, T., Hosohata, J. & Omoto, T. ERG of form deprivation myopia and drug induced ametropia in chicks. Curr Eye Res 15, 79-86 (1996). [PubMed]
72 Ritchey, E. R. et al. Vision-guided ocular growth in a mutant chicken model with diminished visual acuity. Exp Eye Res 102, 59-69, doi:10.1016/j.exer.2012.07.001 (2012). [PubMed]
73 McBrien, N. A., Moghaddam, H. O., Cottriall, C. L., Leech, E. M. & Cornell, L. M. The effects of blockade of retinal cell action potentials on ocular growth, emmetropization and form deprivation myopia in young chicks. Vision Res 35, 1141-1152, doi:10.1016/0042-6989(94)00237-G (1995). [PubMed]
74 Norton, T. T., Essinger, J. A. & McBrien, N. A. Lid-suture myopia in tree shrews with retinal ganglion cell blockade. Vis Neurosci 11, 143-153, doi:10.1017/S0952523800011184 (1994). [PubMed]
75 Wildsoet, C. & Wallman, J. Choroidal and scleral mechanisms of compensation for spectacle lenses in chicks. Vision Res 35, 1175-1194, doi:10.1016/0042-6989(94)00233-C (1995). [PubMed]
76 Dillingham, C. M., Guggenheim, J. A. & Erichsen, J. T. Disruption of the centrifugal visual system inhibits early eye growth in chicks. Invest Ophthalmol Vis Sci 54, 3632-3643, doi:10.1167/iovs.12-11548 (2013). [PubMed]
77 Owusu-Yaw, V., Kyle, A. L. & Stell, W. K. Effects of lesions of the optic nerve, optic tectum and nervus terminalis on rod precursor proliferation in the goldfish retina. Brain Res 576, 220-230 (1992). [PubMed]
78 Avellaneda-Chevrier, V. K., Wang, X., Hooper, M. L. & Chauhan, B. C. The retino–retinal projection: Tracing retinal ganglion cells projecting to the contralateral retina. Neuroscience Letters 591, 105-109, doi:http://dx.doi.org/10.1016/j.neulet.2015.02.033 (2015). [PubMed]
79 Nadal-Nicolas, F. M. et al. Retino-retinal projection in juvenile and young adult rats and mice. Exp Eye Res 134, 47-52, doi:10.1016/j.exer.2015.03.015 (2015). [PubMed]
80 Kolb, H. Roles of Amacrine Cells. Webvision: The Organization of the Retina and Visual System, (Online) http://webvision.med.utah.edu (2012).
81 Masland, R. H. Cell populations of the retina: the Proctor lecture. Invest Ophthalmol Vis Sci 52, 4581-4591, doi:10.1167/iovs.10-7083 (2011). [PubMed]
82 Masland, R. H. The neuronal organization of the retina. Neuron 76, 266-280, doi:10.1016/j.neuron.2012.10.002 (2012). [PubMed]
83 Famiglietti, E. V. Dendritic co-stratification of ON and ON-OFF directionally selective ganglion cells with starburst amacrine cells in rabbit retina. The Journal of comparative neurology 324, 322-335, doi:10.1002/cne.903240303 (1992). [PubMed]
84 Gollisch, T. & Meister, M. Eye smarter than scientists believed: neural computations in circuits of the retina. Neuron 65, 150-164, doi:10.1016/j.neuron.2009.12.009 (2010). [PubMed]
85 Bitzer, M., Kovacs, B., Feldkaemper, M. & Schaeffel, F. Effects of muscarinic antagonists on ZENK expression in the chicken retina. Exp Eye Res 82, 379-388, doi:10.1016/j.exer.2005.07.010 (2006). [PubMed]
86 Feldkaemper, M. P. & Schaeffel, F. Evidence for a potential role of glucagon during eye growth regulation in chicks. Vis Neurosci 19, 755-766 (2002). [PubMed]
87 Lan, W., Yang, Z., Feldkaemper, M. & Schaeffel, F. Changes in dopamine and ZENK during suppression of myopia in chicks by intense illuminance. Exp Eye Res 145, 118-124, doi:10.1016/j.exer.2015.11.018 (2015). [PubMed]
88 Rohrer, B., Iuvone, P. M. & Stell, W. K. Stimulation of dopaminergic amacrine cells by stroboscopic illumination or fibroblast growth factor (bFGF, FGF-2) injections: possible roles in prevention of form-deprivation myopia in the chick. Brain Res 686, 169-181, doi:10.1016/0006-8993(95)00370-6 (1995). [PubMed]
89 Ganesan, P. & Wildsoet, C. F. Pharmaceutical intervention for myopia control. Expert Rev Ophthalmol 5, 759-787, doi:10.1586/eop.10.67 (2010). [PubMed]
90 Bitzer, M. & Schaeffel, F. Effects of quisqualic acid on retinal ZENK expression induced by imposed defocus in the chick eye. Optom Vis Sci 81, 127-136 (2004). [PubMed]
91 Wildsoet, C. F. & Pettigrew, J. D. Kainic acid-induced eye enlargement in chickens: differential effects on anterior and posterior segments. Invest Ophthalmol Vis Sci 29, 311-319 (1988). [PubMed]
92 Li, X. X., Schaeffel, F., Kohler, K. & Zrenner, E. Dose-dependent effects of 6-hydroxy dopamine on deprivation myopia, electroretinograms, and dopaminergic amacrine cells in chickens. Vis Neurosci 9, 483-492, doi:10.1017/S0952523800011287 (1992). [PubMed]
93 McBrien, N. A., Moghaddam, H. O. & Reeder, A. P. Atropine reduces experimental myopia and eye enlargement via a nonaccommodative mechanism. Invest Ophthalmol Vis Sci 34, 205-215, doi:10.1016/j.exer.2006.09.019 (1993). [PubMed]
94 Leech, E. M., Cottriall, C. L. & McBrien, N. A. Pirenzepine prevents form deprivation myopia in a dose dependent manner. Ophthalmic Physiol Opt 15, 351-356, doi:10.1046/j.1475-1313.1995.9500074n.x (1995). [PubMed]
95 Chia, A. et al. Atropine for the treatment of childhood myopia: safety and efficacy of 0.5%, 0.1%, and 0.01% doses (Atropine for the Treatment of Myopia 2). Ophthalmology 119, 347-354, doi:10.1016/j.ophtha.2011.07.031 (2012). [PubMed]
96 Chua, W. H. et al. Atropine for the treatment of childhood myopia. Ophthalmology 113, 2285-2291, doi:10.1016/j.ophtha.2006.05.062 (2006). [PubMed]
97 Tan, D. T., Lam, D. S., Chua, W. H., Shu-Ping, D. F. & Crockett, R. S. One-year multicenter, double-masked, placebo-controlled, parallel safety and efficacy study of 2% pirenzepine ophthalmic gel in children with myopia. Ophthalmology 112, 84-91, doi:10.1016/j.ophtha.2004.06.038 (2005). [PubMed]
98 Raviola, E. & Wiesel, T. N. An animal model of myopia. N Engl J Med 312, 1609-1615, doi:10.1056/nejm198506203122505 (1985). [PubMed]
99 Tigges, M. et al. Effects of muscarinic cholinergic receptor antagonists on postnatal eye growth of rhesus monkeys. Optom Vis Sci 76, 397-407, doi:10.1097/00006324-199906000-00020 (1999). [PubMed]
100 Mckanna, J. A. & Casagrande, V. A. in Third International Conference on Myopia Copenhagen, August 24–27, 1980 (eds H. C. Fledelius, P. H. Alsbirk, & E. Goldschmidt) 187-192 (Springer Netherlands, 1981). [Springer]
101 Arumugam, B. & McBrien, N. A. Muscarinic antagonist control of myopia: evidence for M4 and M1 receptor-based pathways in the inhibition of experimentally-induced axial myopia in the tree shrew. Invest Ophthalmol Vis Sci 53, 5827-5837, doi:10.1167/iovs.12-9943 (2012). [PubMed]
102 Cottriall, C. L. & McBrien, N. A. The M1 muscarinic antagonist pirenzepine reduces myopia and eye enlargement in the tree shrew. Invest Ophthalmol Vis Sci 37, 1368-1379 (1996). [PubMed]
103 Stone, R. A., Lin, T. & Laties, A. M. Muscarinic antagonist effects on experimental chick myopia. Exp Eye Res Vol. 52 755-758 (1991). [PubMed]
104 Luft, W. A., Ming, Y. & Stell, W. K. Variable effects of previously untested muscarinic receptor antagonists on experimental myopia. Invest Ophthalmol Vis Sci 44, 1330-1338, doi:10.1167/iovs.02-0796 (2003). [PubMed]
105 Metlapally, S. & McBrien, N. A. The effect of pirenzepine on positive- and negative-lens-induced refractive error and ocular growth in chicks. Invest Ophthalmol Vis Sci 51, 5438-5444, doi:10.1167/iovs.09-4431 (2010). [PubMed]
106 Bartlett, J. D. et al. A tolerability study of pirenzepine ophthalmic gel in myopic children. J Ocul Pharmacol Ther 19, 271-279, doi:10.1089/108076803321908392 (2003). [PubMed]
107 Cottriall, C. L., McBrien, N. A., Annies, R. & Leech, E. M. Prevention of form-deprivation myopia with pirenzepine: a study of drug delivery and distribution. Ophthalmic Physiol Opt 19, 327-335, doi:10.1046/j.1475-1313.1999.00438.x (1999). [PubMed]
108 Wildsoet, C. Neural pathways subserving negative lens-induced emmetropization in chicks-insights from selective lesions of the optic nerve and ciliary nerve. Curr Eye Res 27, 371-385, doi:10.1076/ceyr.27.6.371.18188 (2003). [PubMed]
109 Barathi, V. A., Weon, S. R. & Beuerman, R. W. Expression of muscarinic receptors in human and mouse sclera and their role in the regulation of scleral fibroblasts proliferation. Mol Vis 15, 1277-1293 (2009). [PubMed]
110 Fischer, A. J., McKinnon, L. A., Nathanson, N. M. & Stell, W. K. Identification and localization of muscarinic acetylcholine receptors in the ocular tissues of the chick. J Comp Neurol 392, 273-284, doi:10.1002/(SICI)1096-9861(19980316)392:3%3C273::AID-CNE1%3E3.0.CO;2-Z (1998). [PubMed]
111 McBrien, N. A., Jobling, A. I., Truong, H. T., Cottriall, C. L. & Gentle, A. Expression of muscarinic receptor subtypes in tree shrew ocular tissues and their regulation during the development of myopia. Mol Vis 15, 464-475 (2009). [PubMed]
112 Strang, C. E., Renna, J. M., Amthor, F. R. & Keyser, K. T. Muscarinic acetylcholine receptor localization and activation effects on ganglion response properties. Invest Ophthalmol Vis Sci 51, 2778-2789, doi:10.1167/iovs.09-4771 (2010). [PubMed]
113 Tietje, K. M. & Nathanson, N. M. Embryonic chick heart expresses multiple muscarinic acetylcholine receptor subtypes. Isolation and characterization of a gene encoding a novel m2 muscarinic acetylcholine receptor with high affinity for pirenzepine. J Biol Chem 266, 17382-17387 (1991). [PubMed]
114 McBrien, N. A., Stell, W. K. & Carr, B. How does atropine exert its anti-myopia effects? Ophthalmic Physiol Opt 33, 373-378, doi:10.1111/opo.12052 (2013). [PubMed]
115 Iribarren, R. Tropicamide and myopia progression. Ophthalmology 115, 1103-1104, doi:10.1016/j.ophtha.2007.11.018 (2008). [PubMed]
116 Siatkowski, R. M. et al. Safety and efficacy of 2% pirenzepine ophthalmic gel in children with myopia: a 1-year, multicenter, double-masked, placebo-controlled parallel study. Arch Ophthalmol 122, 1667-1674, doi:10.1001/archopht.122.11.1667 (2004). [PubMed]
117 Siatkowski, R. M. et al. Two-year multicenter, randomized, double-masked, placebo-controlled, parallel safety and efficacy study of 2% pirenzepine ophthalmic gel in children with myopia. J aapos 12, 332-339, doi:10.1016/j.jaapos.2007.10.014 (2008). [PubMed]
118 Cottriall, C. L., Truong, H. T. & McBrien, N. A. Inhibition of myopia development in chicks using himbacine: a role for M(4) receptors? Neuroreport 12, 2453-2456, doi:10.1097/00001756-200108080-00033 (2001). [PubMed]
119 Vessey, K. A., Cottriall, C. L. & McBrien, N. A. Muscarinic receptor protein expression in the ocular tissues of the chick during normal and myopic eye development. Brain Res Dev Brain Res 135, 79-86, doi:10.1016/S0165-3806(02)00309-7 (2002). [PubMed]
120 Fischer, A. J., Miethke, P., Morgan, I. G. & Stell, W. K. Cholinergic amacrine cells are not required for the progression and atropine-mediated suppression of form-deprivation myopia. Brain Res 794, 48-60, doi:10.1016/S0006-8993(98)00188-7 (1998). [PubMed]
121 Nickla, D. L., Zhu, X. & Wallman, J. Effects of muscarinic agents on chick choroids in intact eyes and eyecups: evidence for a muscarinic mechanism in choroidal thinning. Ophthalmic Physiol Opt 33, 245-256, doi:10.1111/opo.12054 (2013). [PubMed]
122 McBrien, N. A., Arumugam, B., Gentle, A., Chow, A. & Sahebjada, S. The M4 muscarinic antagonist MT-3 inhibits myopia in chick: evidence for site of action. Ophthalmic Physiol Opt 31, 529-539, doi:10.1111/j.1475-1313.2011.00841.x (2011). [PubMed]
123 Carr, B. J. Molecular Targets of Atropine- and Mamba Toxin 3-Mediated Inhibition of Form-Deprivation Myopia in the Chick: A Case of Mistaken Receptor Identity? PhD thesis, University of Calgary, (2017). [Univ. of Calgary]
124 Nickla, D. L., Yusupova, Y. & Totonelly, K. The Muscarinic Antagonist MT3 Distinguishes Between Form Deprivation- and Negative Lens-Induced Myopia in Chicks. Curr Eye Res 40, 962-967, doi:10.3109/02713683.2014.967870 (2015). [PubMed]
125 Potter, L. T. Snake toxins that bind specifically to individual subtypes of muscarinic receptors. Life Sci 68, 2541-2547 (2001). [PubMed]
126 Blanchet, G. et al. Polypharmacology profiles and phylogenetic analysis of three-finger toxins from mamba venom: case of aminergic toxins. Biochimie 103, 109-117, doi:10.1016/j.biochi.2014.04.009 (2014). [PubMed]
127 Nareoja, K. et al. Adrenoceptor activity of muscarinic toxins identified from mamba venoms. Br J Pharmacol 164, 538-550, doi:10.1111/j.1476-5381.2011.01468.x (2011). [PubMed]
128 McDougal, D. H. & Gamlin, P. D. Autonomic control of the eye. Compr Physiol 5, 439-473, doi:10.1002/cphy.c140014 (2015). [PubMed]
129 Woldemussie, E., Wijono, M. & Pow, D. Localization of alpha 2 receptors in ocular tissues. Vis Neurosci 24, 745-756, doi:10.1017/s0952523807070605 (2007). [PubMed]
130 Ruuskanen, J. O., Peitsaro, N., Kaslin, J. V., Panula, P. & Scheinin, M. Expression and function of alpha-adrenoceptors in zebrafish: drug effects, mRNA and receptor distributions. J Neurochem 94, 1559-1569, doi:10.1111/j.1471-4159.2005.03305.x (2005). [PubMed]
131 Iuvone, P. M. & Rauch, A. L. Alpha 2-adrenergic receptors influence tyrosine hydroxylase activity in retinal dopamine neurons. Life Sci 33, 2455-2463 (1983). [PubMed]
132 Iuvone, P. M. Regulation of retinal dopamine biosynthesis and tyrosine hydroxylase activity by light. Fed Proc 43, 2709-2713 (1984). [PubMed]
133 Nguyen-Legros, J., Martin-Martinelli, E., Simon, A., Denoroy, L. & Vigny, A. Co-localization of tyrosine hydroxylase and phenylethanolamine-N-methyltransferase immunoreactivity in the rat retina: a re-examination using double labeling on semi-thin sections. Exp Eye Res 43, 575-584 (1986). [PubMed]
134 Zhou, X., Pardue, M. T., Iuvone, P. M. & Qu, J. Dopamine signaling and myopia development: What are the key challenges. Prog Retin Eye Res, doi:10.1016/j.preteyeres.2017.06.003 (2017). [PubMed]
135 DeVries, S. H. & Schwartz, E. A. Modulation of an electrical synapse between solitary pairs of catfish horizontal cells by dopamine and second messengers. J Physiol 414, 351-375, doi:10.1113/jphysiol.1989.sp017692 (1989). [PubMed]
136 Rohrer, B., Spira, A. W. & Stell, W. K. Apomorphine blocks form-deprivation myopia in chickens by a dopamine D2-receptor mechanism acting in retina or pigmented epithelium. Vis Neurosci 10, 447-453, doi:10.1017/S0952523800004673 (1993). [PubMed]
137 Seko, Y., Tanaka, Y. & Tokoro, T. Apomorphine inhibits the growth-stimulating effect of retinal pigment epithelium on scleral cells in vitro. Cell Biochem Funct 15, 191-196, doi:10.1002/(sici)1099-0844(199709)15:3<191::aid-cbf738>3.0.co;2-2 (1997). [PubMed]
138 Weiss, S. & Schaeffel, F. Diurnal growth rhythms in the chicken eye: relation to myopia development and retinal dopamine levels. J Comp Physiol A 172, 263-270 (1993). [PubMed]
139 Nickla, D. L. & Totonelly, K. Dopamine antagonists and brief vision distinguish lens-induced- and form-deprivation-induced myopia. Exp Eye Res 93, 782-785, doi:10.1016/j.exer.2011.08.001 (2011). [PubMed]
140 Iuvone, P. M., Tigges, M., Stone, R. A., Lambert, S. & Laties, A. M. Effects of apomorphine, a dopamine receptor agonist, on ocular refraction and axial elongation in a primate model of myopia. Invest Ophthalmol Vis Sci 32, 1674-1677 (1991). [PubMed]
141 Schwahn, H. N., Kaymak, H. & Schaeffel, F. Effects of atropine on refractive development, dopamine release, and slow retinal potentials in the chick. Vis Neurosci 17, 165-176, doi:10.1017/S0952523800171184 (2000). [PubMed]
142 Schmid, K. L. & Wildsoet, C. F. Inhibitory effects of apomorphine and atropine and their combination on myopia in chicks. Optom Vis Sci 81, 137-147, doi:10.1097/00006324-200402000-00012 (2004). [PubMed]
143 Stone, R. A., Lin, T., Laties, A. M. & Iuvone, P. M. Retinal dopamine and form-deprivation myopia. Proc Natl Acad Sci U S A 86, 704-706, doi:10.1073/pnas.86.2.704 (1989). [PubMed]
144 Schaeffel, F., Bartmann, M., Hagel, G. & Zrenner, E. Studies on the role of the retinal dopamine/melatonin system in experimental refractive errors in chickens. Vision Res 35, 1247-1264, doi:10.1016/0042-6989(94)00221-7 (1995). [PubMed]
145 McCarthy, C. S., Megaw, P., Devadas, M. & Morgan, I. G. Dopaminergic agents affect the ability of brief periods of normal vision to prevent form-deprivation myopia. Exp Eye Res 84, 100-107, doi:10.1016/j.exer.2006.09.018 (2007). [PubMed]
146 Cottriall, C. L., Brew, J., Vessey, K. A. & McBrien, N. A. Diisopropylfluorophosphate alters retinal neurotransmitter levels and reduces experimentally-induced myopia. Naunyn Schmiedebergs Arch Pharmacol 364, 372-382, doi:10.1007/s002100100460 (2001). [PubMed]
147 Arumugam, B. & McBrien, N. The D2 Antagonist Spiperone Prevents Muscarinic Antagonist Control of Experimentally-Induced Myopia in Chick. IOVS; ARVO E-Abstract 51, 1195 (2010). [IOVS]
148 Arumugam, B. & McBrien, N. Spiperone Prevents M1 Muscarinic Antagonist Inhibition of Myopia in Tree shrew. IOVS; ARVO E-Abstract 53, 3431 (2012). [IOVS]
149 Moinul, P., Cheng, E. & Stell, W. K. Nitric Oxide Prevents Form-Deprivation Myopia in Chicks Without Requiring a Dopamine D2-Receptor Relay. IOVS; ARVO E-Abstract 53, 3434 (2012). [IOVS]
150 Elphick, M. R. Localization of nitric oxide synthase using NADPH diaphorase histochemistry. Methods Mol Biol 72, 153-158, doi:10.1385/0-89603-394-5:153 (1997). [PubMed]
151 Fischer, A. J. & Stell, W. K. Nitric oxide synthase-containing cells in the retina, pigmented epithelium, choroid, and sclera of the chick eye. J Comp Neurol 405, 1-14, doi:10.1002/(SICI)1096-9861(19990301)405:1%3C1::AID-CNE1%3E3.0.CO;2-U (1999). [PubMed]
152 Wilson, M., Nacsa, N., Hart, N. S., Weller, C. & Vaney, D. I. Regional distribution of nitrergic neurons in the inner retina of the chicken. Vis Neurosci 28, 205-220, doi:10.1017/s0952523811000083 (2011). [PubMed]
153 Eldred, W. D. & Blute, T. A. Imaging of nitric oxide in the retina. Vision Res 45, 3469-3486, doi:10.1016/j.visres.2005.07.033 (2005). [PubMed]
154 Tekmen-Clark, M. & Gleason, E. Nitric oxide production and the expression of two nitric oxide synthases in the avian retina. Vis Neurosci 30, 91-103, doi:10.1017/s0952523813000126 (2013). [PubMed]
155 Blom, J., Giove, T., Deshpande, M. & Eldred, W. D. Characterization of nitric oxide signaling pathways in the mouse retina. J Comp Neurol 520, 4204-4217, doi:10.1002/cne.23148 (2012). [PubMed]
156 Nickla, D. L. & Wildsoet, C. F. The effect of the nonspecific nitric oxide synthase inhibitor NG-nitro-L-arginine methyl ester on the choroidal compensatory response to myopic defocus in chickens. Optom Vis Sci 81, 111-118, doi:10.1097/00006324-200402000-00009 (2004). [PubMed]
157 Nickla, D. L., Damyanova, P. & Lytle, G. Inhibiting the neuronal isoform of nitric oxide synthase has similar effects on the compensatory choroidal and axial responses to myopic defocus in chicks as does the non-specific inhibitor L-NAME. Exp Eye Res 88, 1092-1099, doi:10.1016/j.exer.2009.01.012 (2009). [PubMed]
158 Nickla, D. L., Wilken, E., Lytle, G., Yom, S. & Mertz, J. Inhibiting the transient choroidal thickening response using the nitric oxide synthase inhibitor l-NAME prevents the ameliorative effects of visual experience on ocular growth in two different visual paradigms. Exp Eye Res 83, 456-464, doi:10.1016/j.exer.2006.01.029 (2006). [PubMed]
159 Chakraborty, R. et al. Pharmacological inhibition of nitric oxide increases myopia susceptibility in mice. IOVS; ARVO E-Abstract 57, 4742 (2016).
160 Noll, G. N., Billek, M., Pietruck, C. & Schmidt, K. F. Inhibition of nitric oxide synthase alters light responses and dark voltage of amphibian photoreceptors. Neuropharmacology 33, 1407-1412, doi:10.1016/0028-3908(94)90042-6 (1994). [PubMed]
161 Sato, M., Ohtsuka, T. & Stell, W. K. Endogenous nitric oxide enhances the light-response of cones during light-adaptation in the rat retina. Vision Res 51, 131-137, doi:10.1016/j.visres.2010.10.011 (2011). [PubMed]
162 Shiells, R. & Falk, G. Retinal on-bipolar cells contain a nitric oxide-sensitive guanylate cyclase. Neuroreport 3, 845-848, doi:10.1097/00001756-199210000-00006 (1992). [PubMed]
163 Tsuyama, Y., Noll, G. N. & Schmidt, K. F. L-arginine and nicotinamide adenine dinucleotide phosphate alter dark voltage and accelerate light response recovery in isolated retinal rods of the frog (Rana temporaria). Neurosci Lett 149, 95-98, doi:10.1016/0304-3940(93)90356-P (1993). [PubMed]
164 Wellard, J. W. & Morgan, I. G. Nitric oxide donors mimic the effects of light on photoreceptor melatonin synthesis. Aust N Z J Ophthalmol 24, 61-63, doi:10.1111/j.1442-9071.1996.tb00998.x (1996). [PubMed]
165 Shi, Q. Mechanisms of Adaptation to Mean Light Intensity in the Chick Retina PhD thesis, University of Calgary, (2014). [Univ. of Calgary]
166 Bloomfield, S. A. & Volgyi, B. The diverse functional roles and regulation of neuronal gap junctions in the retina. Nat Rev Neurosci 10, 495-506, doi:10.1038/nrn2636 (2009). [PubMed]
167 Teves, M., Shi, H., Eng, D. & Stell, W. K. The role of cell-cell coupling in myopia development and light adaptation. IOVS; ARVO E-Abstract 55, 3036 (2014). [IOVS]
168 Trier, K., Munk Ribel-Madsen, S., Cui, D. & Brogger Christensen, S. Systemic 7-methylxanthine in retarding axial eye growth and myopia progression: a 36-month pilot study. J Ocul Biol Dis Infor 1, 85-93, doi:10.1007/s12177-008-9013-3 (2008). [PubMed]
169 Cui, D. et al. Effects of 7-methylxanthine on the sclera in form deprivation myopia in guinea pigs. Acta Ophthalmol 89, 328-334, doi:10.1111/j.1755-3768.2009.01688.x (2011). [PubMed]
170 Nie, H. H. et al. Effects of 7-methylxanthine on form-deprivation myopia in pigmented rabbits. Int J Ophthalmol 5, 133-137, doi:10.3980/j.issn.2222-3959.2012.02.03 (2012). [PubMed]
171 Carr, B. J. & Stell, W. K. Alpha2-Adrenergic Agonists Inhibit Form-Deprivation Myopia (FDM) in the Chick. IOVS; ARVO E-Abstract 57, 4738 (2016). [IOVS]
172 Cantor, L. B. Brimonidine in the treatment of glaucoma and ocular hypertension. Ther Clin Risk Manag 2, 337-346 (2006). [PubMed]
173 Carr, B. J. & Stell, W. K. Alpha2-Adrenergic Agonists Inhibit Form-Deprivation Myopia (FDM) in the Chick. IOVS; ARVO E-Abstract 57, 4738 (2016). [PubMed]
174 Stone, R. A. et al. GABA, experimental myopia, and ocular growth in chick. Invest Ophthalmol Vis Sci 44, 3933-3946 (2003). [PubMed]
175 Cheng, Z. Y., Wang, X. P., Schmid, K. L. & Han, X. G. Inhibition of form-deprivation myopia by a GABAAOr receptor antagonist, (1,2,5,6-tetrahydropyridin-4-yl) methylphosphinic acid (TPMPA), in guinea pigs. Graefes Arch Clin Exp Ophthalmol 252, 1939-1946, doi:10.1007/s00417-014-2765-5 (2014). [PubMed]
176 Cheng, Z. Y. et al. GABAB receptor antagonist CGP46381 inhibits form-deprivation myopia development in guinea pigs. Biomed Res Int 2015, 207312, doi:10.1155/2015/207312 (2015). [PubMed]
177 Chebib, M. et al. Novel, potent, and selective GABAC antagonists inhibit myopia development and facilitate learning and memory. J Pharmacol Exp Ther 328, 448-457, doi:10.1124/jpet.108.146464 (2009). [PubMed]
178 Schmid, K. L., Strasberg, G., Rayner, C. L. & Hartfield, P. J. The effects and interactions of GABAergic and dopaminergic agents in the prevention of form deprivation myopia by brief periods of normal vision. Exp Eye Res 110, 88-95, doi:10.1016/j.exer.2013.02.017 (2013). [PubMed]
179 Christian, P. G., Harkin, D. G. & Schmid, K. L. GABAergic agents modify the response of chick scleral fibroblasts to myopic and hyperopic eye cup tissues. Curr Eye Res 39, 172-187, doi:10.3109/02713683.2013.834941 (2014). [PubMed]
180 Rohrer, B., Tao, J. & Stell, W. K. Basic fibroblast growth factor, its high- and low-affinity receptors, and their relationship to form-deprivation myopia in the chick. Neuroscience 79, 775-787 (1997). [PubMed]
181 Chen, B. Y., Wang, C. Y., Chen, W. Y. & Ma, J. X. Altered TGF-beta2 and bFGF expression in scleral desmocytes from an experimentally-induced myopia guinea pig model. Graefes Arch Clin Exp Ophthalmol 251, 1133-1144, doi:10.1007/s00417-013-2269-8 (2013). [PubMed]
182 Zhang, D. & Eldred, W. D. Colocalization of enkephalin-, glucagon-, and corticotropin-releasing factor-like immunoreactivity in GABAergic amacrine cells in turtle retina. Brain Res 596, 46-57 (1992). [PubMed]
183 Fischer, A. J., Skorupa, D., Schonberg, D. L. & Walton, N. A. Characterization of glucagon-expressing neurons in the chicken retina. J Comp Neurol 496, 479-494, doi:10.1002/cne.20937 (2006). [PubMed]
184 Ashby, R., Kozulin, P., Megaw, P. L. & Morgan, I. G. Alterations in ZENK and glucagon RNA transcript expression during increased ocular growth in chickens. Mol Vis 16, 639-649 (2010). [PubMed]
185 Vessey, K. A., Lencses, K. A., Rushforth, D. A., Hruby, V. J. & Stell, W. K. Glucagon receptor agonists and antagonists affect the growth of the chick eye: a role for glucagonergic regulation of emmetropization? Invest Ophthalmol Vis Sci 46, 3922-3931, doi:10.1167/iovs.04-1026 (2005). [PubMed]
186 Stell, W. K., Lencses, K. A., Ahm, J. M. & Hruby, V. J. Glucagon synthesizing amacrine cells mediate choroidal compensation for plus-defocus in the chick eye (abstract). European Journal of Neuroscience 12, 487 (2000).
187 Feldkaemper, M. P., Neacsu, I. & Schaeffel, F. Insulin acts as a powerful stimulator of axial myopia in chicks. Invest Ophthalmol Vis Sci 50, 13-23, doi:10.1167/iovs.08-1702 (2009). [PubMed]
188 Zhong, X., Ge, J., 3rd, S. E. L. & Stell, W. K. Selective activation of a subpopulation of GABAergic amacrine cells by focused and defocused images in Macaque retina (abstract). European Retina Meeting, Frankfurt, Germany (2007).
189 Zhong, X., Ge, J., Smith, E. L., 3rd & Stell, W. K. Image defocus modulates activity of bipolar and amacrine cells in macaque retina. Invest Ophthalmol Vis Sci 45, 2065-2074 (2004). [PubMed]
190 McFadden, S. A., Howlett, M. H. & Mertz, J. R. Retinoic acid signals the direction of ocular elongation in the guinea pig eye. Vision Res 44, 643-653, doi:10.1016/j.visres.2003.11.002 (2004). [PubMed]
191 Mertz, J. R. & Wallman, J. Choroidal retinoic acid synthesis: a possible mediator between refractive error and compensatory eye growth. Exp Eye Res 70, 519-527, doi:10.1006/exer.1999.0813 (2000). [PubMed]
192 Seko, Y., Shimokawa, H. & Tokoro, T. In vivo and in vitro association of retinoic acid with form-deprivation myopia in the chick. Exp Eye Res 63, 443-452, doi:10.1006/exer.1996.0134 (1996). [PubMed]
193 Seko, Y., Shimizu, M. & Tokoro, T. Retinoic acid increases in the retina of the chick with form deprivation myopia. Ophthalmic Res 30, 361-367, doi:10.1159/000055496 (1998). [PubMed]
194 Bitzer, M., Feldkaemper, M. & Schaeffel, F. Visually induced changes in components of the retinoic acid system in fundal layers of the chick. Exp Eye Res 70, 97-106, doi:10.1006/exer.1999.0762 (2000). [PubMed]
195 Troilo, D., Nickla, D. L., Mertz, J. R. & Summers Rada, J. A. Change in the synthesis rates of ocular retinoic acid and scleral glycosaminoglycan during experimentally altered eye growth in marmosets. Invest Ophthalmol Vis Sci 47, 1768-1777, doi:10.1167/iovs.05-0298 (2006). [PubMed]
196 Weiler, R., Pottek, M., He, S. & Vaney, D. I. Modulation of coupling between retinal horizontal cells by retinoic acid and endogenous dopamine. Brain Res Brain Res Rev 32, 121-129 (2000). [PubMed]
197 Zhang, D. Q. & McMahon, D. G. Direct gating by retinoic acid of retinal electrical synapses. Proc Natl Acad Sci U S A 97, 14754-14759, doi:10.1073/pnas.010325897 (2000). [PubMed]
198 Wallman, J. et al. Moving the retina: choroidal modulation of refractive state. Vision Res 35, 37-50 (1995). [PubMed]
199 Nickla, D. L., Totonelly, K. & Dhillon, B. Dopaminergic agonists that result in ocular growth inhibition also elicit transient increases in choroidal thickness in chicks. Exp Eye Res 91, 715-720, doi:10.1016/j.exer.2010.08.021 (2010). [PubMed]
200 Nickla, D. L. & Totonelly, K. Choroidal thickness predicts ocular growth in normal chicks but not in eyes with experimentally altered growth. Clin Exp Optom 98, 564-570, doi:10.1111/cxo.12317 (2015). [PubMed]
201 Nickla, D. L. & Wallman, J. The multifunctional choroid. Prog Retin Eye Res 29, 144-168, doi:10.1016/j.preteyeres.2009.12.002 (2010). [PubMed]
202 Summers, J. A. The choroid as a sclera growth regulator. Exp Eye Res 114, 120-127, doi:10.1016/j.exer.2013.03.008 (2013). [PubMed]
203 Bedrossian, R. H. The effect of atropine on myopia. Ophthalmology 86, 713-719 (1979). [PubMed]
204 Tong, L. et al. Atropine for the treatment of childhood myopia: effect on myopia progression after cessation of atropine. Ophthalmology 116, 572-579, doi:10.1016/j.ophtha.2008.10.020 (2009). [PubMed]
205 Chia, A. et al. Atropine for the treatment of childhood myopia: changes after stopping atropine 0.01%, 0.1% and 0.5%. Am J Ophthalmol 157, 451-457.e451, doi:10.1016/j.ajo.2013.09.020 (2014). [PubMed]
206 Chia, A., Lu, Q. S. & Tan, D. Five-Year Clinical Trial on Atropine for the Treatment of Myopia 2: Myopia Control with Atropine 0.01% Eyedrops. Ophthalmology 123, 391-399, doi:10.1016/j.ophtha.2015.07.004 (2016). [PubMed]
207 Xie, P. & Guo, X. Chinese Experiences on Orthokeratology. Eye Contact Lens 42, 43-47, doi:10.1097/icl.0000000000000190 (2016). [PubMed]
208 Cho, P., Cheung, S. W. & Edwards, M. The longitudinal orthokeratology research in children (LORIC) in Hong Kong: a pilot study on refractive changes and myopic control. Curr Eye Res 30, 71-80 (2005). [PubMed]
209 Cho, P. & Cheung, S. W. Retardation of myopia in Orthokeratology (ROMIO) study: a 2-year randomized clinical trial. Invest Ophthalmol Vis Sci 53, 7077-7085, doi:10.1167/iovs.12-10565 (2012). [PubMed]
210 Gifford, P. & Gifford, K. L. The Future of Myopia Control Contact Lenses. Optom Vis Sci 93, 336-343, doi:10.1097/opx.0000000000000762 (2016). [PubMed]
211 Loh, K. L., Lu, Q., Tan, D. & Chia, A. Risk factors for progressive myopia in the atropine therapy for myopia study. Am J Ophthalmol 159, 945-949, doi:10.1016/j.ajo.2015.01.029 (2015). [PubMed]
212 Liu, Y. M. & Xie, P. The Safety of Orthokeratology–A Systematic Review. Eye Contact Lens 42, 35-42, doi:10.1097/icl.0000000000000219 (2016). [PubMed]
213 Kinoshita, N., Konno, Y., Hamada, N. & Kakehashi, A. Suppressive effect of combined treatment of orthokeratology and 0.01% atropine instillation on axial length elongation in childhood myopia. IOVS; ARVO E-Abstract 57, 2386 (2017).
214 Tosini, G., Ferguson, I. & Tsubota, K. Effects of blue light on the circadian system and eye physiology. Mol Vis 22, 61-72 (2016). [PubMed]
About the authors
Brittany Carr graduated with Honours from the Bachelor of Health Sciences program at the University of Calgary in 2011, and earned her PhD in Neuroscience at the Cumming School of Medicine at the University of Calgary, Alberta, Canada, in April 2017. Mentored by Dr. William K. Stell and Dr. Morley D. Hollenberg, her doctorate research was concerned with investigating the signalling mechanisms involved in atropine-mediated inhibition of myopia. By investigating the pharmacological behavior of atropine and other myopia-inhibiting muscarinic acetylcholine receptor (mAChR) antagonists, she successfully demonstrated that myopia inhibition by atropine may not be controlled by mAChRs, but instead some other unknown target receptor(s) or pathway(s), possibly involving alpha2A-adrenoceptors. She is currently a postdoctoral fellow, studying retinal degeneration at the University of British Columbia Centre for Macular Research (Vancouver, BC, Canada).
Bill Stell has been Professor of Cell Biology and Anatomy in the Cumming School of Medicine, University of Calgary, since 1980. He graduated from Swarthmore College with High Honors in Zoology in 1961, and received his Ph.D. in Anatomy in 1966 and M.D. in 1967, both from the University of Chicago. Subsequently he was at the NINDS, NIH, in the laboratories of Drs. Thomas G. Smith, Jr., K.C. Richardson, Henry G. Wagner, and M.G.F. Fuortes. In 1972 he joined the faculty of the Jules Stein Eye Institute, UCLA for 8 years, and finally found a home in Neuroscience at the University of Calgary, Alberta. Originally known for pioneering studies in EM of Golgi-impregnated neurons in fish retina, more recently Bill became interested in the retinal origins of myopia. Since then, he – with outstanding PhD. Students such as Dr. Carr – has made many and varied contributions to understanding the role(s) of the retina in causing and preventing myopia.